safelix1260
05.09.2019 •
Chemistry
The thallium (present as tl2so4) in a 9.486-g pesticide sample was precipitated as thallium(i) iodide. calculate the mass percent of tl2so4 in the sample if 0.1824 g of tli was recovered.
Solved
Show answers
More tips
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry What would your weight on the moon be? *this is your answer for your mass on the moon not mine!...
- C Chemistry The ph scale runs from 0 to 14, and is an indication of how acidic or basic a solution is. a ph close to zero indicates a(n) solution, and a ph near 14 indicates a(n) solution....
- C Chemistry A chemist must dilute 10.3mL of 4.00M aqueous sodium chloride NaCl solution until the concentration falls to 3.00M . He ll do this by adding distilled water to the solution until it...
- C Chemistry What is the amount of heat, in calories, given off from a 5 g piece of aluminum when it cools from 80°C to 20°C? The specific heat capacity of aluminum is 0.215 cal/g∙°C. Show your...
- C Chemistry A 25.0 mL aliquot of 0.0630 M EDTA was added to a 30.0 mL solution containing an unknown concentration of V3+. All of the V3+ present in the solution formed a complex with EDTA, leaving...
- C Chemistry Fossil fuels contain energy that originally came froma.tidal forces.b.earth’s core.c.the sun.d.dinosaurs....
- C Chemistry Calculate the number of s atoms in 0.600 mole of s....
- C Chemistry When in everyday life is evaporation used to separate solutions?...
- S Spanish Que se ignifica los instantes son granos de melancolia ...
- B Biology The diagram below shows three families. Which set of parents has the highest fitness? A- the parents of the single childB- the parents of two childrenC- the parents of the big family...

Ответ:
The mass percentage of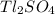 is 5.86%
is 5.86%
Explanation:
To calculate the mass percentage of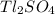 in the sample it is necessary to know the mass of the solute (
in the sample it is necessary to know the mass of the solute (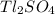 in this case), and the mass of the solution (pesticide sample, whose mass is explicit in the letter of the problem).
in this case), and the mass of the solution (pesticide sample, whose mass is explicit in the letter of the problem).
To calculate the mass of the solute, we must take the mass of the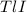 precipitate. We can establish a relation between the mass of
precipitate. We can establish a relation between the mass of 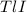 and
and 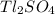 using the stoichiometry of the compounds:
using the stoichiometry of the compounds:
Since for every mole of Tl in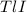 there are two moles of Tl in
there are two moles of Tl in 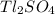 , we have:
, we have:
Using the molar mass of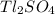 we have:
we have:
Finally, we can use the mass percentage formula:
Ответ:
The answer to your question is below
Explanation:
Noble gases has their levels of energy completed so they can not react with another element, they can not interchange electrons .
electronic configuration
He 1 s ²
Ne 1s² 2s² 2p⁶
Ar [Ne] 3s² 3p⁶
Kr [Ar] 3d¹⁰ 4s² 4p⁶
Xe [Kr] 4d¹⁰ 5s² 5p⁶
Rn [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p⁶
In the electronic configurations we can see that Noble gases have 8 electrons in their outermost shell, so they do not need to react to be stable, they are stable.