alyssamaize
17.12.2019 •
Chemistry
To begin the experiment, 1.11g of methane ch4 is burned in a bomb
calorimeter containing 1000 grams of water. the initial temperature of water is 24.85oc. the specific heat of water is 4.184 j/g oc. the heat capacity of the calorimeter is 695 j/ oc . after the reaction the final temperature of the water is 35.65oc.
2. calculate the change in temperature, δt
3. using the formula qwater = m • c • δt ,calculate the heat absorbed by the water.
4. using the formula qcal = ccal • δt ,calculate the heat absorbed by the calorimeter.
5. the total heat absorbed by the water and the calorimeter can be calculated
by adding the heat calculated in steps 3 and 4. the amount of heat released
by the reaction is equal to the amount of heat absorbed with the negative
sign as this is an exothermic reaction. using the formula ∆h = -(qcal qwater + ) ,
calculate the total heat of combustion.
6. evaluate the information contained in this calculation and complete the
following sentence:
this calculation shows that burning grams of methane [takes
in/gives off] energy.
7. the molar mass of methane is 16.04 g/mol. calculate the number of moles
of methane burned in the experiment.
8. what is the experimental molar heat of combustion?
9. the accepted value for the heat of combustion of methane is -890 kj/mol.
explain why the experimental data might differ from the theoretical value.
10.give the formula theoretical value - experimental value % error = ×100
theoretical value , calculate
the percent error for the experiment.
with any of
Solved
Show answers
More tips
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry (a) at 800 k the equilibrium constant for i2(g) 2 i(g) is kc = 3.1 10-5. if an equilibrium mixture in a 10.0-l vessel contains 3.25 10-2 g of i(g), how many grams...
- C Chemistry Adrug user who is experiencing a flashback has probably used . a. speed b. lsd c. downers...
- C Chemistry 9.05 mol of oxygen gas to g of oxygen gas...
- C Chemistry Chegg what is the purpose of cyclohexane reagent prodcut reducing agent axeotropic solvent...
- C Chemistry What is the reaction catalyzed by catalase? a. and what is the substrate chegg...
- C Chemistry Which combination of solutions of hcl and naoh would produce the largest ∆t?...
- C Chemistry A solution is 1.405 m nacl. what is the molarity of the solution if the density is 1.10 g/ml?...
- C Chemistry the shape of the meniscus of water in a glass tube is different from that of mercury in a glass tube. why?...
- C Chemistry Explain the effect of naf on the rate of reaction? why? be specific chegg...
- C Chemistry the chemical formula for chlorophyll is c₅₅h₇₂mgn₄o₅. given the molar mass of the compound is 893.49 g/mol, what quantity in moles of chlorophyll are there in 5.989...

Ответ:
Explanation:
Initial temperature of water = 24.85°C
Final temperature of water = 35.65°C
Mass of water = 1000 g
The specific heat of water ,c = 4.184 J/g °C.
The heat capacity of the calorimeter = 695 J/ °C
= 695 J/ °C
Change in temperature
Heat absorbed by the water =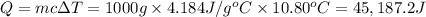
Heat absorbed by the calorimeter =
Total heat combustion =
Heat absorbed by the water + Heat absorbed by the calorirmeter
=(-45,187.2 J)+ (-7,506 J) = -52,693.2 J
1.11 grams of methane gives off 52,693.2 J.
Number of moles methane burned in an experiment =
Experimental molar heat of combustion =
Negative sign heat is released during burning of methane.
The experimental data might differ from the theoretical value because while performing an experiment there are certain factors which affects the the observations and experimental procedure which results in differ value experimental value from theoretical value.
The percentage error in the experiment:
The percent error for the experiment is 14.4 %.
Ответ:
The correct option is a.
Explanation:
125 kPa
125kpa - 2x 4x x
Total pressure after reaction = 176 kPa
125 kPa - 2x + 4x + x = 176 kPa
x = 17
125 kpa - 2x = 125 kPa - 2(17) = 91 kPa
Initial pressure of the dinitrogen pentoxide ,(at t=0) =
Final pressure of the dinitrogen pentoxide, (at t = t) = P = 91 kPa
The rate constant is =
It will take 113 minutes for the total pressure to reach 176 kPa.