shanekamayo1120
26.12.2019 •
Chemistry
Two atoms have the same number of protons but different numbers of neutrons. what can you conclude about these two atoms? a-they are the same element b-they are the same isotope c-they have the same mass d-thay have the same charge.
Solved
Show answers
More tips
- C Computers and Internet How to Reinstall Windows: A Detailed Guide for Beginners...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Chemistry
- C Chemistry 3. Katelyn is working on a project about classifying cells as either prokaryotic or eukaryotic. She is observing a cell under an electron microscope that does not have a cell...
- C Chemistry The percent composition of a compound was found to be 63.5% silver, 8.2% nitrogen, and 28.3% oxygen. Determine the compound s empirical formula....
- C Chemistry Sir Isaac Newton proposed that movements of the bodies in our Solar System are controlled by an invisible universal force called...
- C Chemistry NaBr (aq) + H3PO4 (aq) →...
- C Chemistry Warm air rises at the equator and cold air sinks at the poles creating A. tornadoes B. hurricanes C. convection currents D. the coriolis effect...
- C Chemistry How much energy will be required to heat 5.00 g of iron 20.0 C? The specific heat of iron is 0.45 J/gC....
- C Chemistry What determines the average kinetic energy of the particles in a gas?...
- C Chemistry Explain how photosynthesis and cellular respiration are cycles. The reactants of photosynthesis are and they react with light to make the products and . The reactants of cellular...
- C Chemistry How fast is 12 meters in 4 seconds...
- C Chemistry A raindrop has a mass of 0.050 g. How many moles of water does a raindrop contain?...

Ответ:
A-they are the same element
Explanation: An element is characterized by its atomic number or the number of protons.
Two atoms having the same number of protons but different numbers of neutrons will have same atomic number but different mass number.Thus they are isotopes of the same element.
As Atomic number = Number of protons
Mass number = number of protons + number of neutrons
For example: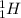 and
and 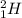 are both isotopes of Hydrogen with same number of protons but different number of neutrons.
are both isotopes of Hydrogen with same number of protons but different number of neutrons.
Ответ: