swagjlove32
05.08.2019 •
Chemistry
Two solutions are prepared using the same solute: solution a.0.27 g of the solute dissolves in 27.4 g of t-butanol solution b.0.23 g of the solute dissolves in 24.8 g of cyclohexane which solution has the greatest freezing point change?
Solved
Show answers
More tips
- L Leisure and Entertainment How to Learn to Draw Graffiti: Tips for Beginners...
- P Photography and Videography How to Choose a Digital Camera?...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Someone plz help I Don’t know I would do something like this and I really need to get it done...
- M Mathematics What is the lateral surface area of the prism...
- M Mathematics Multiply four and eight tenths multiplied by three. thirteen and one tenth thirteen and four tenths fourteen and two tenths fourteen and four tenths...

Ответ:
ΔTf = (Kf)(m)
where ΔTf is freezing point depression, Kf is the molal freezing point depression and m is the molality of the solute. Using the givens above and the equation,
a. Let x be the molar mass of the solute. The Kf of t-butanol is 9.1 °C/m.
ΔTf = (9.1)(0.27/x)(1/0.0274)
ΔTf = 89.67/x
b. Let x be the molar mass of the same solute. Kf value of 20.1 °C/m
ΔTf = (20.1)(0.23/x)(1/0.0248)
ΔTf = 186.4/x
The answer would be letter B.
Ответ:
First balance the equation: you get 3Fe + 4H2O = Fe3O4 +4H2O
Then set up the equations according to the spidey map below.
x 10^25 molec. H2O/ 1 x x x
Multiply the top all together, then divide by the whole bottom half.
154.4 g H2
136 g Fe /1 x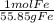 x
x 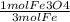 x
x 
Multiply the top all together, then divide by the whole bottom half.
4.95 x 10^20 Formula units (fu) Fe3O4
8.25 x 10^23 molec. Fe/ 1 x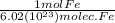 x
x 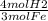
Multiply the top all together, then divide by the whole bottom half.
1.83 mol H2