kaylailkanic1487
20.04.2020 •
Chemistry
Typical audience for a vaudeville show
Solved
Show answers
More tips
- F Food and Cooking Learn How to Make Ice Cream at Home - Step by Step Guide...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry Which of the following factors affect the vapor pressure of a liquid? Check all that apply. View Available Hint(s) Check all that apply. atmospheric pressure surface...
- P Physics the surface tension of liquids depend on the density acceleration of gravity and the height use the method of dimensions to derive the relationship between these quantities...
- M Mathematics What is the expression for 6-2x+5+4x...
- M Mathematics (-3,8) (2, - 11) Х. у. X2 Y -19 m = Y₂ - Y, X - X -11 - 8 2 - (-3) 5...
- S Social Studies Sociologists refer to observable facts or events that involve human society as a. a sociological perspective. b. social phenomena. C. a social interaction. d. sociological...

Ответ:
intended for all-male audience
Explanation:
Ответ:
half life =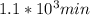 ,
, 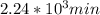 are taken for the concentration to decrease to 25% and
are taken for the concentration to decrease to 25% and 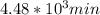 for the concentration to decrease to 6.25% .
for the concentration to decrease to 6.25% .
Explanation: The given information says, the reaction is first order with respect to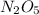 . For first order reaction, rate constant and half life are related to each other as:
. For first order reaction, rate constant and half life are related to each other as:
half life =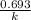
(where k stands for rate constant)
Value of k is given as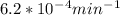
half life =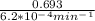
half life = 1117.74 min or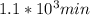
Let's say the initial concentration is 100. It asks to calculate the time taken to decrease the concentration to 25% which will be 25 as we have taken the initial concentration 100.
The equation that we use is:
Where,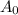 is initial and A is remaining concentration or amounts. k is rate constant and t is the time. Let's plug in the values and do calculations for t.
is initial and A is remaining concentration or amounts. k is rate constant and t is the time. Let's plug in the values and do calculations for t.
t = 2241.94 min or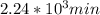
We can calculate the time when the concentration decreases to 6.25% of its initial value same as we did for the above.
t = 4483.87 min or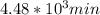
So, the answers for all the three parts are: half life =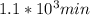 ,
, 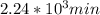 are taken for the concentration to decrease to 25% and
are taken for the concentration to decrease to 25% and 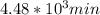 for the concentration to decrease to 6.25% .
for the concentration to decrease to 6.25% .