GabbyGood1072
27.09.2021 •
Chemistry
Use dimensional analysis and the given densities to perform
the following conversions,
a) What is the volume, in milliliters, of a sample of boron with a mass of 24.8 g?
The density of boron is 2.34 g/cm'.
b) What is the volume, in cubic centimeters, of a 7.62 g sample of mercury?
The density of mercury is 13.5 g/cm,
rocoding archlem (#hy annlying the density equation
Solved
Show answers
More tips
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- M Mathematics PLEASE HELP ME ILL MARK YOU BRAINLIEST :(...
- M Mathematics A farmer has 20 bins of apples. Each bir has 25 red apples and 30 green apples. Which of the following expressions can be used to find the total number of apples in all the bins?...
- M Mathematics What are two numbers that round to 130 when rounded to the nearest tenth...

Ответ:
The molarity of the solution is 1.854 mol/L
Explanation:
The weight of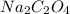 present in 131.64mL of 2.7 M solution:
present in 131.64mL of 2.7 M solution:
Hence, the molarity of the solution is 1.854 mol/L