use the information provided to determine δh°rxn for the following reaction: ch4(g) 4 cl2(g) → ccl4(g) 4 hcl(g) δh°rxn = ?
Solved
Show answers
More tips
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
- A Auto and Moto What is the Average Lifespan of an Engine in a Car?...
- H Health and Medicine What You Need to Know About Nasal Congestion in Infants: Causes, Symptoms, and Treatment...
- S Science and Technology Discovering the Anatomy of an LCD TV Screen...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- F Food and Cooking Why Doesn t the Confirmation Link Come to Email?...
- F Food and Cooking Deflope: What is it and how does it work?...
Answers on questions: Chemistry
- C Chemistry 2. A chemical reaction occurs when substances interact and their atoms O A. disappear B. change their size. C. become new atoms. O D. rearrange...
- C Chemistry The compound whose molecules have the highest average kinetic energy is?...
- C Chemistry Pls help mmmmmmmmmmmmmmmmmmmmmmmmmmmmm...
- C Chemistry Orange light has a wavelength of 585 nm. calculate the frequency and energy of the orange light....
- C Chemistry Amixture of helium, neon and argon gases have a total pressure of 790 mmhg. if there is 118.5 mmhg of argon, 474 mmhg of helium, what is the partial pressure of the neon gas?...
- C Chemistry What is the molar mass of a pure gaseous compound having a density of 2.95 g/l at 32⁰c and 860 mm hg? (1 atm = 760 mm hg)?...
- C Chemistry How many acidic hydrogens in acetic acid?...
- C Chemistry Atank holding 7.80 l of gas is at a pressure of 9.80 atm. if the temperature and number of gas particles do not change, what is the new pressure at a volume of 5.3 l? *show work*...
- C Chemistry Ill give brainliest This is super easy im just lazy what s the name of this compound N2P9...
- C Chemistry Please help this is due in 30 minutes!!...

Ответ:
The given question is incomplete.. The complete question is :
Consider the following reaction:
At equilibrium, a 4.50 L container has 2.6 g of carbon, CO2 at a partial pressure of 0.0020 atm, and a total pressure of 0.572 atm. Calculate KP for this reaction at 725oC.
162.45
Explanation:
Equilibrium pressure of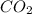 = 0.0020 atm
= 0.0020 atm
Total pressure at equilibrium = 0.572 atm
Equilibrium pressure of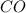 = Total pressure at equilibrium - Equilibrium pressure of
= Total pressure at equilibrium - Equilibrium pressure of 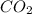 = (0.572- 0.0020) = 0.570 atm
= (0.572- 0.0020) = 0.570 atm
The given balanced equilibrium reaction is,
At eqm 0 0.0020 0.570
The expression for equilibrium constant for this reaction will be,
Now put all the given values in this expression, we get :
By solving we get :
Thus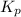 for this reaction at
for this reaction at 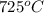 is 162.45
is 162.45