Orange light has a wavelength of 585 nm. calculate the frequency and energy of the orange light.
Solved
Show answers
More tips
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- W Work and Career Мерчендайзинг – все, что нужно знать...
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
Answers on questions: Chemistry
- C Chemistry Amole of a gas at stp has a volume of 22.4 l. if the volume increases to 29.1 l and the pressure is changed to 813 torr, calculate the new temperature....
- C Chemistry How does percolation filter water?...
- M Mathematics Lars deposited $50 into a savings account. according to the rule of 72, what interest rate will cause his money to double in approximately 29 years?...
- S Social Studies Sociologists use the phrase racial literacy to refer to the fact that...

Ответ:
3.4 x 10⁻¹⁹J
Explanation:
Given parameters:
Wavelength = 585nm
Unknown:
Frequency and energy of the wave = ?
Solution:
The frequency of the wave can be derived from;
C = f x wavelength
c is the speed of light = 3 x 10⁸m/s
f is the frequency
Insert the parameters and solve;
3 x 10⁸ = f x 585 x 10⁻⁹
f =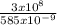
f = 5.13 x 10⁻³ x 10¹⁷ = 5.13 x 10¹⁴m⁻¹
the energy of the wave;
E = hf
h is the planck's constant;
E = 6.63 x 10⁻³⁴ x 5.13 x 10¹⁴ = 34.01 x 10⁻²⁰J or 3.4 x 10⁻¹⁹J
Ответ: