tabbydory3366
24.02.2020 •
Chemistry
Use the standard reaction enthalpies given below to determine ΔH°rxn for the following reactionP4(g) + 10 Cl2(g) → 4PCl5(s) ΔH°rxn = ?Given:PCl5(s) → PCl3(g) + Cl2(g) ΔH°rxn = +157 kJP4(g) + 6 Cl2(g) → 4 PCl3(g) ΔH°rxn = -1207 kJ
Solved
Show answers
More tips
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- D Dating, Love, Relationships Why do we feel shame?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
Answers on questions: Chemistry
- C Chemistry Which word could mean an arrow? ( --- )...
- C Chemistry Write the chemichal formulae of the following compounds.1) Ammonium chloride2) calcium hydroxide...
- C Chemistry hat colour is thymol blue in a 4.0 x 10-4 mol/L solution of potassium hydroxide? What colour is indigo carmine?...
- C Chemistry three chemicals, p , q and r were each dissolved in water. the table shows some of the reaction of these solutions...
- C Chemistry In halogens, fluorine has fewer electrons and a weaker dispersion force as compared to iodine. which statement is true regarding fluorine and iodine? a. fluorine is a gas and...
- C Chemistry According to the transparency how is nitrogen returned to the atmosphere...
- C Chemistry What is the mole fraction of ethanol in the vapor phase over an ethanolmethanol solution that has a mole fraction of ethanol of 0.30 in the liquid phase? The vapor pressures...
- C Chemistry What is the molality of a solution prepared by dissolving 75.8 g of ethylene glycol, HOCH2CH2OH, in 1.55 L of water? Assume the density of water is 1.00 g/mL....
- C Chemistry The release of heat energy is a common indicator of a chemical reaction true or false...
- C Chemistry Assume that a raindrop has a volume of 3.48 cm3. if you had an avogadro’s number of raindrops that just filled a cubic box. what would be the edge of the length of the box measured...

Ответ:
Therefore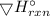 = -1835 KJ
= -1835 KJ
Explanation:
Enthalpy is denoted by H.
Enthalpy: Total heat change in a chemical reaction is called enthalpy.
The change of entalpy of a reaction is denoted by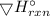
Hass's Law:The change in enthalpy of any process can be determined by calculating the sum of change in enthalpy of each of the steps involved in the process.
g= gas
S= solid
P₄(g)+10Cl₂(g)→ 4Cl₅(s)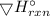 =?
=?
PCl₅(s)→ PCl₃(g)+Cl₂(g) .......(1)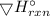 = +157KJ
= +157KJ
P₄(g)+6Cl₂(g)→ 4PCl₃(g).............(2)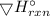 = -1207 KJ
= -1207 KJ
If we flip a reaction the value of enthalpy will be change positive to negative or nagative to positive but the numerical value will be remain same.
We need rearrange the equation (1) because in the required equation Cl₂ is on the left side. So we flip the first equation.
PCl₃(g)+Cl₂(g)→PCl₅(s)......(3)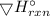 = -157KJ
= -157KJ
Multiplying 4 with equation (3)
4 PCl₃(g)+4Cl₂(g)→4PCl₅(s)......(4)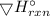 =4×( -157)KJ= -628 KJ
=4×( -157)KJ= -628 KJ
Adding equation (2) and (4) we get
P₄(g)+6Cl₂(g)+4 PCl₃(g)+4Cl₂(g)→4PCl₃(g)+4PCl₅(s)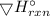 =( -1207-628)KJ
=( -1207-628)KJ
⇒P₄(g)+10Cl₂(g)→4PCl₃(g)-4PCl₃(g)+4PCl₅(s)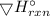 = - 1835KJ
= - 1835KJ
⇒P₄(g)+10Cl₂(g)→ 4Cl₅(s)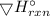 = -1835 KJ
= -1835 KJ
Therefore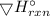 = -1835 KJ
= -1835 KJ
Ответ:
The air pressure at sea level is about 14.7 pounds per square inch (psi). For every foot above sea level, the air pressure drops about 0.0005263 psi. According to this information, at about what height above sea level is the air pressure 13.2 psi?