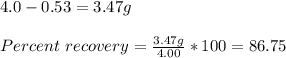Vertical columns on the periodic table are known as
and horizontal rows are called
O periods, groups
groups, periods
O metals, nonmetals
O metalloids, metals
Solved
Show answers
More tips
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- H History Which two european countries had established the most colonies in africa by the end of the 1800s?...
- M Mathematics Consider the Following equation. Determine the missing coordinate in the orderd pair (-4,?) so that it will satisfy the given equation. (-4,______)...
- B Business Acustomer has a group of inside sales cloud users that need to see tracking information at the contact or lead level, but do not need to send an email. which two recommendations should...
- M Mathematics Brainliest and five stars to the right answer...
- C Chemistry How do the isotopes hydrogen-2 and hydrogen-3 differ? (Hint: determine protons, neutrons, and electrons for each isotope.) a. Hydrogen-3 has one more neutron than hydrogen-2. neutron...

Ответ:
Maximum theoretical percent recovery of acetanilide is 86.8%
Explanation:
From the given information:
Solubility of acetanilide in 100 ml water at 100 C = 5.5 g
Solubility of acetanilide in 100 ml water at 0 C = 0.53 g
When 4.0 g of acetanilide is dissolved in 100 ml of water, all of it will go into solution. As the solution is cooled to 0 C only 0.53 g of it will dissolve.
Amount of acetanilide undissolved =