Wenzhou prepares 200 ml of a solution of sncl4in which the concentration ofchloride ions is 0.240m.a) what is the molarity of the sncl4solution (i.e. what should the bottle be labeled)? b) what mass of sncl4did wenzhou use?
Solved
Show answers
More tips
- F Food and Cooking Pu-erh Tea: History, Varieties, Benefits, and Risks...
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- F Food and Cooking How to Make Thin Pancakes: Recipe and Tips...
- S Style and Beauty Is Hot Scissor Haircutting Beneficial or Dangerous?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking Is Bacon Good for You?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
Answers on questions: Chemistry
- C Chemistry Why phenol is more acidic than alcohol?...
- C Chemistry Conclude your research on humans and humpback whales by answering these questions: What conclusion can you draw about the evolution of humans and humpback whales? Did your research...
- C Chemistry How many milliliters of a 2.5 M MgCl, solution contain 17.5 g MgCl2? (Use M as a conversion factor)...
- C Chemistry 50 POINTS Which is a mixture whose solute particles are individual ions or molecules? solvent suspension colloid solution...
- C Chemistry Which is a characteristic of a scientific claim? A.it is supported by a controlled experiment with multiple trials that can be repeated by others B.it is another way to describe...
- C Chemistry What has 3 sigma bonds 2 pi bonds and has Dipole and London Dispersion Forces...
- M Mathematics Marcus paid $40 to buy a potato cannon, a cylinder that shoots potatoes hundreds of feet. He was willing to pay $45. When Marcus s friend Nick learns that Marcus bought a potato...
- C Chemistry Please help need it badly...
- M Mathematics If circle b is similar to circle a, what must exist?...
- M Mathematics Why does someone named amphitrite keep deleting my answers it s getting kind of annoying...

Ответ:
a) 0.06 M
b) 3.13 grams.
Explanation:
Molarity of a solution is defined as the number of moles of solute dissolved per Liter of the solution.
Moles of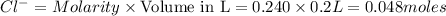
The balanced reaction for dissociation will be:
Thus for 4 moles of , there is 1 mole of
, there is 1 mole of 
Thus moles of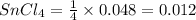
Molarity of
Thus the molarity of the solution = 0.06 M
solution = 0.06 M
b) Mass of =
= 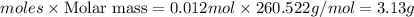
Thus mass of Wenzhou use is 3.13 grams.
Wenzhou use is 3.13 grams.
Ответ: