andrwisawesome0
27.09.2020 •
Chemistry
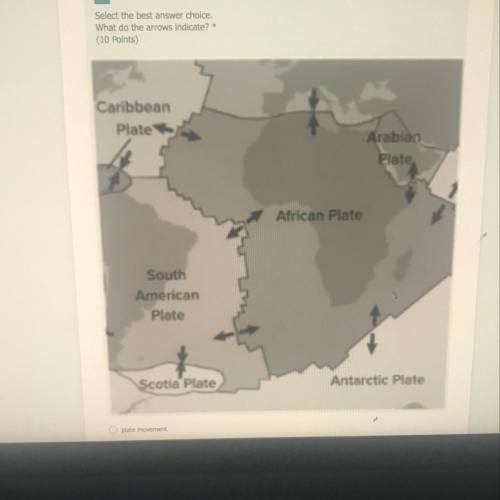
What do the arrows indicate ? A)plate movement. B)ocean flow. C)mid ocean ridge. D)continental drift.
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of Under Eye Bags?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- P Physics A92-kg skier is sliding down a ski slope that makes an angle of 30 degrees above the horizontal direction. the coefficient of kinetic friction between the skis and the snow is...
- M Mathematics stickers mermaid with the same ratio of length to width. a sticker one inch wide has a link of 2 in. what is the length of stickers of the 5 in wide? complete the ratio table...
- E English Read the sentence. He worked feverishly, barely ever looking up as his pencil flew up and down the pages of his notebook, and within an hour had solved every problem in every...
- M Mathematics Help I need this done ASAP anyone know how to solve this...

Ответ:
55.0125 grams NO2
Explanation:
So we have 80 grams of O2, first convert to moles
80 g O2 * 1 mol/32 g O2 = 2.5 mol O2
Next use the mole ratio of O2 to NO2
2.5 mol O2* 1 mol NO2/2 mol NO2=1.25 mol NO2
Since the question is asking how many grams, convert NO2 to grams
1.25 mol NO2 * 44.01 NO2/1 mol NO2= 55.0125 grams