velazquezemmy45
11.06.2020 •
Chemistry
What does Avogadro's law say about a gas atSTP? A; 1 L of any gas contains 22.4 moles at STP B; 1 L of any gas has a pressure of 22.4 atm at STP C; All gases are at 22.4 atm at STP D; 1 mole of any gas has a volume of 22.4 L at STP
Solved
Show answers
More tips
- F Food and Cooking How to Cook a Steak with Mustard Sauce? What Kind of Meat to Choose?...
- P Photography and Videography What is lens calibration and why is it needed?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- P Physics The second law of themodynamics says that whenever energy is converted from one form to another in a physical or chemical change a. heat is absorbed b. light is produced c. the...
- M Mathematics Which number line shows the solutions to x 5...
- C Chemistry Determine the moles of H+ reacting with the metal based on the following experimental data. 10.00 mL of 1.00 M HCl solution is added to a sample of Mg metal. The reaction goes to...

Ответ:
1 mole of any gas has a volume of 22.4 L at STP
Explanation:
According to avogadro's law, 1 mole of every substance occupies 22.4 L at STP and contains avogadro's number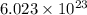 of particles.
of particles.
According to ideal gas equation:
P = pressure of gas = 1 atm (at STP)
V = Volume of gas = ?
n = number of moles = 1
R = gas constant =
T =temperature = (at STP)
(at STP)
Thus 1 mole of every gas occupies 22.4 L at STP
Ответ:
I would use calorimetric to determine the specific heat and I would measure the mass of a sample. I would measure the mass of a sample of the substance. I would heat the substance to a known temperature. I would place the heated substance into a coffee-cup calorimeter containing a known mass of water with a known initial temperature. I would wait for the temperature to equilibrate, then calculate temperature change. I would use the temperature change of water to determine the amount of energy absorbed. I would use the amount of energy lost by substance, mass, and temperature change to calculate specific heat.
Explanation: