KarlaOsorio7779
31.03.2020 •
Chemistry
What happens to boiling point and freezing point of water when solute is added?
1. Bp is raised and Fp is lwoered
2. Bp is lowered and FP is raised
3. Bp is lowered and Fp is lowered
4. Bp is raised and Fp is raised
Solved
Show answers
More tips
- C Computers and Internet Connecting Your TV to Your Computer: Easy and Fast...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- P Physics One beaker contains 100 ml of pure water and second beaker contains 100 ml of seawater. the two beakers are left side by side on a lab bench for one week. at the end of the...
- M Mathematics Jada correctly determines four arithmetic means between −11 and 16. what values does jada determine? jada determines the four arithmetic means to be −5.6 , −0.2 , 5.2, and...
- M Mathematics Avector u has a magnitude of 15 and a direction of 0 degrees. a vector v has a magnitude of 9 and a direction of 35 degrees. find the direction and magnitude of u+v to the...
- M Mathematics I need help ASAP pls...
- C Chemistry When 5 grams of salt dissolve in 30 grams of water, what is the resulting mass of the mixture?...

Ответ:
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.
Explanation:
According to the rules of electron filling in subshells in quantum mechanics , Aufbau's principle states that :
"An electron must be filled in a lower energy state or subshell and then higher. If they have the same energy , the one with lesser principal quantum number will be filled first."
Thus ,
The series of filling goes like:
Thus , the subshells in the given information have 1,3 and 1 orbitals respectively. Thus it has to be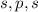 which is possible in two cases :
which is possible in two cases :
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.