Bescobar017
03.05.2021 •
Chemistry
What is an Independent Variable (IV)? *
Solved
Show answers
More tips
- B Business and Finance What is the Difference Between Visa and Visa Gold?...
- A Animals and plants Why do cats go crazy over catnip?...
- W Work and Career Everything You Need to Know About MBA Programs...
- S Sport How to Do Push-ups Correctly?...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
Answers on questions: Chemistry
- C Chemistry Calculate the mass of sodium acetate trihydrate required to make 100. ml of a 0.100 m solution. 0.820 g 1.00 g 1.36 g 1.95 g...
- C Chemistry Ionic bonds contain atoms of bonded with atoms of...
- C Chemistry If I have 2 liters of gas held at a pressure of 78 atm and a temperature of 900 k, what will be the volume of the gas if I decrease the pressure to 45 atm and decrease...
- M Mathematics How to factor x^2-2x-1...
- M Mathematics C=a(w/150) solve for w...
- M Mathematics PLEASE HELP 20 POINTS WILL BE GIVEN WAY...
- S Social Studies Briefly describe the achievements of Pythagoras Euclid Archimedes and Hippocrates...
- M Mathematics Find the area of the polygon. Note that the figure is not drawn to scale. (I THINK I FOUND THE ANSWER BUT HAVE 2 CHECK!)...
- H History What was the pseudonym used for the sixteen anti-federalist papers that were written in response to the Federalist Papers?...
- M Mathematics What is the unknown angle measure in this pattern?...

Ответ:
here's your answer dear friend❤❤
Ответ:
What is the mole fraction of a solution that contains 200 g of magnesium hydroxide in 225 g of water
0.215
Explanation:
Given parameters:
Mass of magnesium hydroxide = 200g
Mass of water = 225g
Unknown:
Mole fraction of solution = ?
Solution:
The mole fraction is a measurement of concentration that expresses the number of moles of solute to that of solution.
Mole fraction =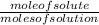
A solution is made up of solute and solvent
Number of moles =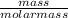
Molar mass of Mg(OH)₂ = 24.3 + 2(16 + 1) = 58.3g/mol
Molar mass of H₂O = 2(1) + 16 = 18g/mol
Number of moles of Mg(OH)₂ =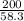 = 3.43moles solute
= 3.43moles solute
Number of moles of H₂O =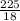 = 12.5moles solvent
= 12.5moles solvent
Mole fraction =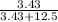 = 0.215
= 0.215