sahaitong814
23.06.2019 •
Chemistry
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Solved
Show answers
More tips
- P Philosophy How did the concept of module arise in computer science?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- H Health and Medicine At What Age Does a Person Stop Growing?...
- F Family and Home How to Choose a Name for Your Baby?...
- F Food and Cooking Discover the Health Benefits of Cedar Nuts...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- F Food and Cooking How to Make Sushi: A Step-by-Step Guide to Perfectly Rolled Delights...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- F Food and Cooking Аэрогриль: Все, что нужно знать для подготовки здоровой и вкусной пищи...
Answers on questions: Chemistry
- S Social Studies 14.285 points SAVE ANSIER QUESTION 2 Qualitative adule Part H A common convention in social science is to set the alpha level at .05 and to reject the null hypothesis if the observed...
- M Mathematics -6 4-5 -3 -2 -1 -9 -8 -7 -6 -5 -4 6 8 9 - 1 -2 3 -4 -5 -6 -7 Jine -8 -9 x-intercept(s): y-intercept(s): Domain: of Use Range:...
- M Mathematics Confusing again ?!?!?!?! Please help I have 5 mins...

Ответ:
The molecular formula for the compound is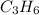
Explanation:
As with all of these problems, we assume 100 g of an unknown compound.
And thus, we determine the elemental composition by the given percentages.
Moles of carbon = 85.64 / 12.011
= 7.13 mol.
Moles of hydrogen = 14.36 / 1.00794
= 14.25 mol.
There are 2 moles of hydrogen per mole of carbon. And thus the empirical formula is CH .
.
And molecular formula = n × (empirical formula)
Thus, 42.08 = n × (12.011 + 2 × 1.00794)
And thus n = 3, and molecular formula =