robert7248
09.01.2020 •
Chemistry
What is the best name for
k[cr(nh3)2cl2so4]
a. potassium (iii).
b. potassium (ii).
c. potassium (iii) sulfate.
d. potassium diamminedichlorochromate(ii) sulfate.
e. potassium (iii).
Solved
Show answers
More tips
- S Sport How to Do a Jumping Split...
- A Animals and plants What Do Terriers Eat?...
- F Food and Cooking Discover the Benefits and Properties of Dates...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- E English Which of the following best states the purpose of Wilson s anti-trust legislation? to encourage free enterprise to protect big business to support workers rights...
- B Biology ASAP NEEd HElp QuIcK One day, you ride your bike to a local bookstore. You purchase a few of your favorite comic books, put them in your backpack, and ride home....
- B Biology An injury to the brain would hamper the sleep-wake cycle...

Ответ:
E. potassium diamminedichlorosulfatochromate (III).
Explanation
Rules for naming of the coordination compounds :
a) The ligand named first then metal.
b) The charge of metal indicated with the Roman numerals and enclosed in the parenthesis after metal name.
c) The ligands are named in alphabetical order before the metal ion.
d) Ligands that include a numerical prefix in the name use the prefixes bis for 2, tris for 3, or tetrakis for 4 to indicate their number.
e) If the complex ion is an anion, we drop the ending of the metal name and add –ate.
In the given coordination compound, Cr is the chromium metal, ,
, 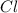 and
and  are the ligands named as ammine, chloro and sulphato.
are the ligands named as ammine, chloro and sulphato.
The charge on![[Cr(NH_3)_2Cl_2SO_4]](/tpl/images/0448/1233/adc39.png) is -1. Ammine is a neutral ligand, chloro is having an oxidation state of -1 and sulphato is having an oxidation state of -2.
is -1. Ammine is a neutral ligand, chloro is having an oxidation state of -1 and sulphato is having an oxidation state of -2.
x+2(0)+2(-1)+1(-2)=-1
x=+3
Hence, the IUPAC name of![K[Cr(NH_3)_2Cl_2SO_4]](/tpl/images/0448/1233/d097e.png) is potassium diaaminedichlorosulphato chromate (III)
is potassium diaaminedichlorosulphato chromate (III)
Ответ: