aliyahlknox881
21.02.2020 •
Chemistry
What is the composition, in atom percent, of an alloy that contains a) 44.5 lbm of silver, b) 84.7 lbm of gold, and c) 7.3 lbm of Cu? The atomic weights for silver, gold, and copper are, respectively, 107.87, 196.97, and 63.55 g/mol.
Solved
Show answers
More tips
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- O Other What is a Disk Emulsifier and How Does it Work?...
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- C Chemistry Lara claims that a water strider will be able to walk on the surface of acetone, which is commonly known as fingernail polish remover. Do you agree or disagree with Lara...
- C Chemistry If the size, mass, and arrangement of atoms affect the density of a substance. How might these factors work together to cause a substance to have a low density?...
- C Chemistry Which of these is a base? a.) ammionia b.) HCI c.) HNO3 d.) vinigar...
- C Chemistry 1-Are natural resources renewable or nonrenewable? 2-what is the only way to stop the depletion of natural resources? 3-What is an example of poor soil management?...
- C Chemistry HELP PLZZ ANSWER THE QUESTION! CORRECT GETS BRAINLIEST...
- C Chemistry What is always true about the total amount of O2 and CO2 in the test tube?...
- C Chemistry Fe + CuSO4 → FeSO4 + Cu What type of reaction is shown above?...
- C Chemistry Which redox reaction would most likely occur if zinc and copper metal were added to a solution that contained zinc and copper ions?...
- C Chemistry Why is the formula for magnesium phosphate not Mg3PO42? Since 3 and 42 have a GCF, the formula needs to be reduced. The correct formula is MgPO13. Since 3 and 42 have...
- E English Outlines should make use of “coordination,” which means that...

Ответ:
The atom percent of silver, gold and copper in the alloy is 43.08 %, 44.93 % and 11.99 % respectively
Explanation:
To convert the given masses into grams, we use the conversion factor:
1 lb = 453.6 grams
To calculate the number of moles, we use the equation:
To calculate the atom percent of substance in sample, we use the equation:
where,
 = Avogadro's number
= Avogadro's number
Moles of Silver:Mass of silver = 44.5 lb = 20185.2 grams
We know that:
Molar mass of silver = 107.87 g/mol
Putting values in equation 1, we get:

Moles of Gold:Mass of gold = 84.7 lb = 38419.9 grams
We know that:
Molar mass of gold = 196.97 g/mol
Putting values in equation 1, we get:

Moles of Copper:Mass of copper = 7.3 lb = 3311.3 grams
We know that:
Molar mass of copper = 63.55 g/mol
Putting values in equation 1, we get:
Total moles of the sample =
For Silver:Moles of silver = 187.12 moles
Total moles = [187.12 + 195.05 + 52.10] = 434.27 moles
Putting values in equation 2, we get:

For Gold:Moles of gold = 195.05 moles
Total moles = [187.12 + 195.05 + 52.10] = 434.27 moles
Putting values in equation 2, we get:
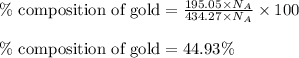
For Copper:Moles of copper = 52.10 moles
Total moles = [187.12 + 195.05 + 52.10] = 434.27 moles
Putting values in equation 2, we get:
Hence, the atom percent of silver, gold and copper in the alloy is 43.08 %, 44.93 % and 11.99 % respectively
Ответ:
420 mmHg x 1 atm/760 mmHg = 0.553 atm
Then use one of the gas laws to solve.
P1V1=P2V2
Plug in the known values and solve for the one remaining.
(2.15atm)(2.00L)=(0.553atm)(V2)
4.3=0.5533V2
V2=4.3/0.5533
V2=7.77 Liters