kakalli9999
02.12.2021 •
Chemistry
What is the density of kcl at 25.00 °c if the edge length of its fcc unit cell is 628 pm?
Solved
Show answers
More tips
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- D Dating, Love, Relationships How Long Can Love Last?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- F Food and Cooking How to Find Your Zip Code?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- S Style and Beauty How to Apply Foundation Correctly?...
Answers on questions: Chemistry
- C Chemistry UestionsThe chicken that Amal ate for lunch atewheat. Wheat is a plant. Draw a food chainshowing how the energy passed from theSun to Amal when he ate the chicken.Draw a food...
- C Chemistry Why are chemical equations balanced? Write an example of balanced chemical equation....
- C Chemistry Draw an example of a buffer in the body....
- C Chemistry Type the correct answer in the box. Use numerals instead of words. Fiona has a lovely flower bed in her yard. She wants to add a fence along one side of the flower bed. The...
- C Chemistry Plz help! :D What element is as hard as magnesium?...
- C Chemistry Identify the product formed from the phosphorylation of adp....
- C Chemistry If a sulfur ion with 16 protons and 17 electrons loses one electron, what is the resulting charge?...
- C Chemistry Metal dissolved into another metal is called...
- C Chemistry A0.100 g sample of a carbon–hydrogen–oxygen compound is combusted in a stream of pure oxygen and produces 0.220 g co2 and 0.239 g h2o. what is the mass percent of hydrogen in...
- C Chemistry This quantity tells me how many grams of a substance is in one mole of a substance: mole ratio molar mass avogadro s number...

Ответ:
The relationship between the equilibrium constant of reaction and the rate constants for the forward
of reaction and the rate constants for the forward  and reverse
and reverse  reactions in a single step reaction is
reactions in a single step reaction is 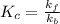
Explanation:
Equilibrium constant is the ratio of product of the concentration of products to the product of concentration of reactants.
For example if the reaction is :
Rate of forward direction=![k_f[X]](/tpl/images/0701/1901/d6b0b.png)
Rate of backward direction=![k_b[Y]](/tpl/images/0701/1901/fc03b.png)
At equilibrium, Rate of forward direction = Rate of backward direction
thus
where
Thus