rachellynn02
28.09.2019 •
Chemistry
What is the hydroxide ion concentration for muriatic acid (has a ph of 2.5)?
Solved
Show answers
More tips
- W Work and Career How to behave at an interview? Tips from an HR specialist...
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry HELPPP!! 8th grade schedule science...
- C Chemistry Why does solid zinc chloride not conduct electricity when solid...
- C Chemistry Explain shear adhesion....
- C Chemistry In an experiment to find the thermal energy transfer of a material the following characteristics were found: Mass = 10 kg Initial Temp = 20 C Final Temp = 40 C C = 1400...
- C Chemistry Define Magnetic force and Magnetic field...
- C Chemistry Points Possible 15youAnswer each of the following questions clearly and thoughtfully.1. Complete the chart below using your households tychart below using your household...
- C Chemistry 4. Label all stereocenters in the following molecules. How many stereoisomers are possible for each molecule? To be sure, draw each proposed steresoisomer, and check to...
- C Chemistry How many grams of NO can be produced from the following reaction if 68.2 grams of NO2 is consumed? 3NO2(g) + H2O(l) → 2HNO3(aq) + NO(g) 1. 133 g 2. 314 g 3. 0.148 g 4....
- C Chemistry 8.50 of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 138./gmol, is burned completely in excess...
- C Chemistry Raw water is treated water that is sent out for use by humans True or False?...

Ответ:
The concentration of the hydroxide ion concentration for muriatic acid is 3.16 * 10^-12 M
Explanation:
Muriatic acid = HCl
pH = 2.5
pH = -log[H+]
pOH = 14 - pH
pOH = 14 - 2.5 = 11.5
pOH = -log [OH-]
11.5 = -log [OH-]
[OH-] = 10^-11.5
[OH-] = 3.16 * 10^-12 M
The concentration of the hydroxide ion concentration for muriatic acid is 3.16 * 10^-12 M
Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of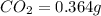
Mass of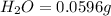
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,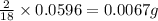 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =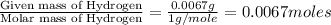 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =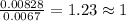
For Hydrogen =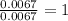
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.