jayleneeeee
01.04.2021 •
Chemistry
What is the kinetic energy of a bicycle with a mass of 14 kg traveling at a velocity of 3
m/s?
joules
Solved
Show answers
More tips
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- C Chemistry what does an energy diagram illustrates identify what the Hell on the energy curve and the positions of the reactants and products represent...
- B Business Martinez Construction, a large firm that has traditionally taken on many different types of projects, has begun to struggle. Many smaller firms in its area have found market sectors...
- E English What is the nomination process at the national party conventions?...
- H History Munch works were well received by the public who understood messages he was trying to convey...
- S Social Studies Although land mines cost about $3, it can cost hundreds of dollars to remove one safely....
- M Mathematics Find equations of the spheres with center (2,-3,6) that touch (a) the xy plane, (b) yz plane, (c) the xz plane....

Ответ:
63 Joule
Solve: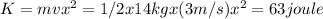
Hope this helped!
Ответ:
a)pH = 4.74
b) pH = 6.75
c) pH = 12.36
Explanation:
In titration of strong base with a weak acid, initially we get salt of the acid and base and due to presence of weak acid in the solution this results into formation of a buffer.
On complete neutralization, the salt undergoes hydrolysis.
After complete neutralization, the solution becomes basic and the pH is due to presence of hydroxide ions.
a) on addition of 10.0 mL of NaOH
The initial moles of acetic acid present = molarity X volume = 0.1 X 0.025 =0.0025
The moles of NaOH added = molarity X volume = 0.125 X 0.01 = 0.00125
The moles of acid left after reaction with base = 0.0025 -0.00125 = 0.00125
the moles of salt formed = 0.00125
the pH of solution will be pH of the buffer formed [pKa of acetic acid =4.74]
b) Addition of 20.0 mL of NaOH
The initial moles of acetic acid present = molarity X volume = 0.1 X 0.025 =0.0025
The moles of NaOH added = molarity X volume = 0.125 X 0.02 = 0.0025
The moles of acid left after reaction with base = 0.0025 -0.0025 = 0
the moles of salt formed = 0.0025
The salt will undergo hydrolysis as:
The Kb will be
[salt] = moles / total volume = 0.0025 / 25+20 = 5.56 X 10⁻⁶ M
Kb = 5.56 X 10⁻¹⁰ M
therefore
5.56 X 10⁻¹⁰ M X 5.56 X 10⁻⁶ M = [OH⁻]²
[OH⁻] = 5.56 X 10⁻⁸ M
pOH = 7.25
pH = 14 -7.25 = 6.75
c) on addition of 30 mL of NaOH 20 mL will be used for complete neutralization and rest 10 mL will be of NaOH only.
moles of NaOH left = molarity X volume = 0.125 X 10 mL = 0.00125
volume of solution = 30 + 25 = 55
the [NaOH] = moles / volume = 0.023 M
pOH = 1.64
pH = 12.36