dbn4everloved8
15.07.2019 •
Chemistry
What is the mass in grams of 1.02 x 1024 atoms manganese (mn)? a. 0.112 x 101 g c. 9.30 x 10-1g b. 0.169 x 101 g d. 9.31 x101 g select the best answer from the choices provided a b c d
Solved
Show answers
More tips
- W Work and Career Мерчендайзинг – все, что нужно знать...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
Answers on questions: Chemistry
- H Health Which of the following type of raw meat is most likely to cause salmonella? a.beef b. chicken c. fish d. lamb...
- S Spanish A que consideras q se debio el crecimiento de la poblacion urbana...
- E English In this excerpt from Jack London s To Build a Fire replace each bolded word with its correct synonym So long as he walked four miles an hour, he pumped that blood,...

Ответ:
Option (d) is the correct answer.
Explanation:
According to Avogadro's number, every 1 mole of a substance contains atoms or particles.
atoms or particles.
Now, it is given that there are atoms of manganese are present. Hence, calculate its number of moles as follows.
atoms of manganese are present. Hence, calculate its number of moles as follows.
No. of moles =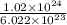
= 1.69 mol
As we know that, number of moles is equal to mass of a substance divided by its molar mass. Since, molar mass of manganese is 54.93 g/mol.
No. of moles =
1.69 mol =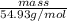
mass = 93 g
Therefore, we can conclude that the mass in grams of atoms of manganese (Mn) are
atoms of manganese (Mn) are 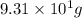 .
.
Ответ: