conroyjoann22
14.04.2020 •
Chemistry
What is the molar mass of Na2Cr2O7?
Solved
Show answers
More tips
- S Science and Technology When do we change our clocks?...
- A Animals and plants How to Teach Your Parrot to Talk?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry Students apply new knowledge to other contexts in which phase?...
- C Chemistry How are the ten ecoregions of Texas determined? What properties make them different from one another?...
- C Chemistry In the reaction 2co ( g) + o2( g) → 2co2( g), what is the ratio of moles of oxygen used to moles of co2produced?...
- C Chemistry Structural formulas are like models because they show _[blank]_. which answer best completes this statement? a condensed way of writing a compound how elements arrange within...
- C Chemistry 14. how would you correctly record the volume in the graduated cylinder below?...
- C Chemistry She dissolves a 10.0mg sample in enough water to make 30.0ml of solution. the osmotic pressure of the solution is 0.340torr at 25c. a). what is the molar mass of the gene fragment?...
- C Chemistry Lexi is researching crime rates. she is trying to make a conclusion about whether the crime rate changes with the population in each county in her state. what is the dependent...
- M Mathematics 4(1-3n)-14 4(2n+3)-9n...
- M Mathematics Expressions. Simplify 2a + 8a...
- M Mathematics PLEASE HELP LAST QUESTION I GIVE THANKS...

Ответ:
The volume of gas is 48.69L.
Explanation:
To calculate the number of moles of gas, we use the equation:
Given mass of water = 36 g
Molar mass of water = 18 g/mol
Putting values in above equation, we get:
Now, to calculate the volume of gas, we use the equation given by ideal gas, which is:
where,
P = pressure of the gas = 100 kPa
V = volume of the gas = ? L
n = Number of moles of gas = 2 mol
R = Gas constant =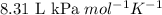
T = temperature of the gas = 293 K
Putting values in above equation, we get:
Hence, the volume of gas is 48.69L.