What is the net ionic equation for the chemical reaction between text
{h}_2 text{so}_4(aq)
h 2 so 4 (aq)start text,
h, end text, start subscript, 2, end subscript, start text, s, o, end text, start subscript, 4, end subscript, (, a, q, )and text
{naoh}(aq)
naoh
(aq)
start text, n, a, o, h, end text, (, a, q, )?
Solved
Show answers
More tips
- G Goods and services Top 5 Best Laundry Detergents for Impeccable Cleanliness of Your Clothes...
- S Society and Politics Как правильно поддерживать температуру в квартире для здоровья...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...

Ответ:
The answer is 0.
Explanation:
Hydrogen ion has a net charge of +1 and sulphate ion has a net charge of -2. Therefore 2moles of Hydrogen ion must react with 1mole of sulphate ion to give H2SO4.
2H+ + SO42- > H2SO4
Ответ:
Explanation:
Given:
Charge = q
Electric field strength =
weight of the droplet = mg
The charge is suspended motionless. This is because the electric force on the charge is balanced by the weight of the droplet.
electric force on charged droplet,
This is balanced by the weight,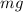
Equating the two: