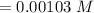dilaydi1212
14.01.2020 •
Chemistry
What is the oxidation half-reaction for mg(s) + zncl2(aq) mgcl2(aq) + zn(s)?
a. zn(s) --> zn^2+ + 2e-
b. mg(s) --> mg2+ + 2e-
c. zn^2+ + 2e- --> zn(s)
d. mg^2+ + 2e- --> mg(s)
Solved
Show answers
More tips
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- B Biology How does the giraffe’s long legs help them survive nature?...
- P Physics Prove that if r is a commutative ring and n = (a) is a finitely generated ideal, where a = \{a_1,\ldots,a_n\} and each a_n is nilpotent, then n is a nilpotent ideal. deduce that if...
- H History Which of the following is not characteristic of the tallmadge amendment? as a group, the southern representatives supported the amendment. children of slaves would be free when they...
- M Mathematics Lollololrgmr4ijngwierjnejgnhwhjvb hreeqhuvcbwfhbqeirfs...
- C Chemistry Mr Smith is changing the oil in his car he dumpster or down the drive to live nearby River how does this affect the animal that live in this habitat...

Ответ:
Ответ:
B)
Explanation: Oxidation is loss of electrons and reduction is gain of electrons. When an element or ion loses electrons then its positive charge increases and when it gains electrons then charge decreases.
In the given equation, on reactant side Mg has zero charge and Zn has +2 charge. On product side, Mg has +2 charge and Zn has zero charge.
Since the charge of Mg is increasing from zero to +2, its oxidation.
We can ignore choice A and C since none of these have Mg. Choice D could also be ignored as it shows reduction of Mg as its changing from +2 to 0 state.
Hence, the correct choice is B as it shows an oxidation of Mg.
B)
Ответ:
The right answer is Option d (0.00103 M).
Explanation:
The given values are:
T = 31.6
e = 487 L mol cm
length, l = 1 cm
As we know,
Absorbance,
By using the Beer's law, we get
⇒
⇒
On substituting the values, we get
⇒
⇒
⇒