annalaurie7
21.01.2020 •
Chemistry
What is the percent yield of a reaction in which 3.8 moles of iron metal react with excess sulfur to produce 230 grams of iron(ii)sulfide? fe + s --> fes
Solved
Show answers
More tips
- H Health and Medicine Where is the appendix located?...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
Answers on questions: Chemistry
- C Chemistry Formula a: ch3 formula b: c2h6 given the two formulas representing the same compound: which statement describes these formulas? (1) formulas a and b are both empirical....
- C Chemistry Which property of a liquid is used when petrol, (gasoline), is pumped from the tank to the engine of a car? *...
- C Chemistry If the mass of reactants equals 100g, then the mass of the products would be equal to 100 g O 50 g O 200 g O 400 g...
- C Chemistry A 1.20g +/- 0.01g sample of hydrated MgCO3 is heated to drive of the water. The dry sample has a mass of 0.58g +/- 0.01g....
- C Chemistry What is it when a cup of ice turns to water...
- C Chemistry Can someone help me with this please...
- C Chemistry A chemical reaction was used to produce 2.95 moles of copper(II) bicarbonate, Cu(HCO3)2. What mass of copper(II) bicarbonate was produced? ___g...
- M Mathematics Which student consumed the most calories? Mario Tim Kim Kia...
- B Biology NEED SERIOUS HELP, i need to know this...
- E English Which choice best describes a difference in how the texts approach the topic of the Oklahoma oil boom? A .Excerpt from Betty Gordon in the Land of Oil talks about...

Ответ:
The answer to your question is 68.8%
Explanation:
Data
Iron = 3.8 moles
Sulfur = excess
FeS = 230 g
Molecular weight FeS = 88 g
Process
Reaction
Fe + S ⇒ FeS
1.- Convert the number of moles of Iron to grams
56 g of Iron -------------- 1 mol
x -------------- 3.8 moles
x = (3.8 x 56) / 1
x = 212.8 g of Iron
2.- Calculate the theoretical production of FeS
56 g of Iron ---------------- 88 g of FeS
212.8 g of iron ----------- x
x = (212.8 x 88) / 56
x = 334.4 g of FeS
3.- Calculate the percent yield
% yield =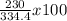
% yield = 68.8
Ответ:
The percent yield of this reaction is 68.9 %
Explanation:
Step 1: Data given
Number of moles of iron = 3.8 moles
Iron reacts with excess of sulfur
230 grams of iron(II)sulfide is produced.
Step 2: The balanced equation
Fe + S → FeS
Step 3: Calculate moles FeS
For 1 mol Fe we need 1 mol S to produce 1 mol FeS
For 3.8 moles Fe we'll have 3.8 moles FeS
Step 4: Calculate mass of FeS
Mass FeS = moles FeS * molar mass FeS
Mass FeS = 3.80 moles * 87.91 g/mol
Mass FeS = 334 grams
Step 5: Calculate % yield
% yield = (actual yield / theoretical yield)*100%
% yield = (230/334) *100%
% yield = 68.9%
The percent yield of this reaction is 68.9 %
Ответ: