What is the vsepr shape of h2co3
Solved
Show answers
More tips
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Chemistry Which event always involves a chemical change?...
- C Chemistry Help and please show the work with it...
- C Chemistry Ernest Rutherford s experiment led him to change the outlook on atom because...
- C Chemistry In which substance are the particles close together and slowly moving past each other? A air Bice C steam D water...
- C Chemistry Can somebody plz help me with number 4...
- C Chemistry I am lustrous and conduct electricity/heat where am I on the periodic table...
- M Mathematics What is the product? 12 64 ION 78 30 CA 32 78 30 0 64 78 30 CAM 32 39 15 6 64...
- M Mathematics An online music store is offering a promotion. When you spend $30 or more, you receive 3 songs for free. You Buy an album for $12 right and solve any quality that represents...
- E Engineering Exhaust gas from a furnace is used to preheat the combustion air supplied to the furnace burners. The gas, which has a flow rate of 15 kg/s and an inlet temperature of 1100 K,...
- C Chemistry Which is a “big idea” for space and time? Forces describe the motion of the universe. Energy can be transferred but not destroyed. The universe consists of matter. The universe...

Ответ:
Answer : The shape of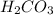 is, trigonal planar.
is, trigonal planar.
Explanation :
Lewis-dot structure : It shows the bonding between the atoms of a molecule.
When we are determining the shape of a molecule then it is important to draw a Lewis-dot structure and for drawing a Lewis-dot structure we should know that how many total number of valance electrons present in a given compound or a molecule.
The given molecule is,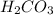
As we know that hydrogen has '1' valence electrons, carbon has '4' valance electrons and oxygen has '6' valence electron.
Therefore, the total number of valence electrons in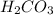 = 2 + 4 + 18 = 24
= 2 + 4 + 18 = 24
Now we have to determine the hybridization of carbon in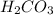
Number of bond pairs = 3
Number of lone pairs = 0
Number of atomic orbitals around carbon atom = 3 + 0 = 3
So, hybridization will be and the shape is, trigonal planar.
and the shape is, trigonal planar.
The Lewis-dot structure is shown below.
Ответ:
Hope this helps! Tbh am not good at explaining. If u have any doubts, ask me!