jayjinks976
22.01.2020 •
Chemistry
What is true about energy in an ordinary chemical reaction?
Solved
Show answers
More tips
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry 5. You add 11.00 mL of 0.800 M NaOH to 50.00 mL of pure water, and to this mixture you then add 3.00 mL of 0.200 M HCl. What will be the pH of the resulting solution...
- C Chemistry The melting of a solid is process because heat energy is the melting of a solid is process because heat energy is endothermic/released endothermic/absorbed exothermic/absorbed...
- C Chemistry An ecosystem is most likely to return to its original condition after which of these happen? a. tall prarie grass burns after being struck by lightning. b. flood waters...
- C Chemistry What can you conclude about the relative concentrations of ch3nh2 and ch3nh3cl in this buffer? for ch3nh2, pkb = 3.36?...
- C Chemistry Which two properties are more typical of molecular compounds than of ionic compounds? 1. they are gases or liquids at room temperature. 2. they have high melting points....
- C Chemistry In a mixture of he, ne, and xe gases with a total pressure of 925 atm, if there is 10.5 g of each gas in the mixture, what is the partial pressure of xe?...
- C Chemistry Explain the connection between your body and supernova...
- C Chemistry Identify the dependent ,independent and controlled variables ? Please help meee I’am stuk in it Helpp!!...
- C Chemistry How many times thus the earth fit inside the sun...
- C Chemistry what precaution will you take when performing an experiment to determine whether glucose is present in different food stuff?...

Ответ:
Ответ:
1) 2.0 g
2) 0 g
3) 4.17 g
4) 2.57 g
Explanation:
First of all, we need to know the compounds and the reaction. The ion carbonate is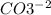 , and the ion nitrate is
, and the ion nitrate is 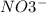 .
.
Sodium is in group 1, so it must lose one electron to be stable, and be the cation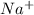 . Silver has only one electron too, so the cation will be
. Silver has only one electron too, so the cation will be 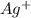 .
.
To form the chemical compounds, first we put the cation, then the anion, and change their charges without the signal:
Sodium carbonate: Na2CO3
Silver nitrate: AgNO3
Silver carbonate: Ag2CO3
Sodium nitrate: NaNO3
The balanced reaction will be:
Na2CO3 + 2 AgNO3 --> Ag2CO3 + 2 NaNO3
Now, we must check the stoichiometry, which will be 1:2:1:2 (always in number of moles)
The question wants to know the mass value, so we need to know the molar mass of these compounds. Checking the periodic table will see that:
Na = 23 g/mol, C = 12 g/mol, N = 14 g/mol, O = 16 g/mol, Ag = 108 g/mol
So the molar mass of the compounds must be:
Na2CO3 = 106 g/mol (2x23 + 12 + 3x16)
AgNO3 = 170 g/mol (108 + 14 + 3x16)
Ag2CO3 = 276 g/mol (2x108 + 12 + 3x16)
NaNO3 = 85 g/mol
We have a mixture of the reactants, so one probably would be in excess, so, first will need to test. Let's do the stoichiometry calculus using silver nitrate as the limit, so:
1 mol of Na2CO3 ---------- 2 mol of AgNO3
106 g ------------------------------ 2x170 = 340 g
x ------------------------------------ 5.14 g
By a simple direct three rule:
340x = 544.84
x = 1.6 g of Na2CO3
That means that for this reaction, we only need 1.6 g of Na2CO3 to react with 5.14 of AgNO3. How we have 3.60 g of Na2CO3, it is on excess, and all the AgNO3 will be consumed.
1) The mass of Na2CO3 that remains after the reaction will be the initial less the mass that reacted:
m = 3.6 - 1. 6 = 2.0 g
2) All the AgNO3 reacted, so there isn't a mass present after the reaction.
m = 0 g
3) Now, doing the stoichiometry calculus between AgNO3 and Ag2CO3
2 moles of AgNO3 ------------- 1 mol of Ag2CO3
2x170 g ------------------------------- 276 g
5.14 g --------------------------------- x
By a simple direct three rule:
340x = 1418.64
x = 4.17 g of Ag2CO3
4) Now, doing the stoichiometry calculus between AgNO3 and NaNO3
2 moles of AgNO3 ----------------------- 2 moles of NaNO3
2x170 g ---------------------------------------- 2x85 g
5.14 g ------------------------------------------- x
By a simple direct three rule:
340x = 873.8
x = 2.57 g