What is true of a covalent bond?
it is the electrostatic attraction between oppositely charged particles
it is the sharing of electrons by overlapping orbitals
it involves the exchange of electrons from one atom to another
it involves the sharing of neutrons between two nuclei
Solved
Show answers
More tips
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to Braid Hair with a Plaiting Machine: Tips and Recommendations...
- C Computers and Internet Where did torrents.ru move to?...
- H Health and Medicine How to Get Pregnant Faster?...
- A Animals and plants Money Tree Care Secrets: How to Keep Your Plant Thriving...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- H Health and Medicine What was the Invention of Viagra?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...

Ответ:
it is the sharing of electrons by overlapping orbitals
Explanation:
Covalent bonds occurs when the reacting elements share their valence electrons in order to attain the duplet or octet configuration of the noble gas.
Ответ:
This reaction is A. Spontaneous at high temperatures, and non-spontaneous at low temperatures.
Explanation:
Both the enthalpy change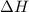 and the entropy change
and the entropy change 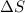 due to this reaction are positive. A chemical reaction will be spontaneous only if the change in its Gibbs Free Energy
due to this reaction are positive. A chemical reaction will be spontaneous only if the change in its Gibbs Free Energy 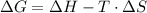 is negative.
is negative. 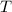 is the absolute temperature in degrees Kelvins.
is the absolute temperature in degrees Kelvins.
Assume that both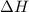 and
and 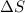 doesn't change much as
doesn't change much as 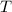 increases. The value of
increases. The value of 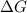 will initially be close to
will initially be close to 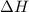 when
when 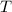 is small. The sign of
is small. The sign of 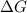 will depends on that of
will depends on that of 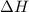 . However,
. However, 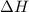 is positive, so at low temperatures
is positive, so at low temperatures 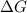 will be positive and the reaction will be non-spontaneous.
will be positive and the reaction will be non-spontaneous.
However, as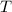 increases, the role of entropy change becomes more significant. The sign of
increases, the role of entropy change becomes more significant. The sign of 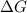 will eventually be the opposite of
will eventually be the opposite of 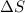 . The value of
. The value of 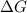 will eventually drop below zero after the value of
will eventually drop below zero after the value of 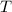 rises above
rises above 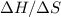 . The reaction will eventually become spontaneous.
. The reaction will eventually become spontaneous.