ftshaechan
20.07.2019 •
Chemistry
What mass of kbr is present in 25 ml of a 0.85 m solution of potassium chloride?
Solved
Show answers
More tips
- F Food and Cooking How to Determine Healthy, Nutritious Food for Yourself?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Chemistry
- C Chemistry What is one of the three parts of cell theory...
- C Chemistry Which is a polar molecule? a) nitrogen trichloride b )sulfur hexachloride c) nitrogen d) carbon disulphide...
- C Chemistry 29. Pure substances may be eitheror compounds *...
- C Chemistry What type of circuit is illustrated a) series circuit b) short circuit c) open circuit d) parallel circuit...
- C Chemistry Are corals minerals? Why?...
- C Chemistry What happens to the atomic radius when an electron is gained? O A. The negative ionic radius is larger than the neutral atomic radius. O B. The negative ionic radius...
- M Mathematics if ▵JKL is an equilateral triangle, JK= 13x+5, KL= 17x-19, & JL=8x+35, find x and the measure of each side....
- M Mathematics Algebra 2 help! Write the Multiplier for the Following: Increase of 12%...
- H History Once a country develops, it has an inclination to grow in life expectancy and A. death rate B. education C. poor people...
- C Chemistry What does it mean to read between the lines?...

Ответ:
Answer : The correct answer for mass of KBr = 2.53 g
Given :
Molarity of KBr solution = 0.85 M
Volume of KBr solution = 25 mL
Converting volume from mL to L ( 1 L = 1000 mL )
Volume of solution = 0.025 L
Mass of KBr = ?
Mass of KBr can be calculated using following steps :
1) To find mole of Kbr :
Mole of KBr can be calculated using molarity .
Molarity : It is defined as mole of solute present in volume of solution in Liter .
It uses unit as M or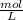
It can be expressed as :
Plugging value of molarity and volume
Multiplying both side by 0.025 L
Mole of KBr = 0.02125
2) To find mass of Kbr :
Mass of Kbr can be calculated using mole . Mole can be expressed as :
Mole of Kbr = 0.02125 mol
Molar mass of KBr = 119.00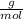
Plugging values in mole formula
Multiplying both side by 119.00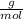
Mass of KBr = 2.53 g
Ответ: