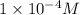nolimitsteve3210
06.01.2022 •
Chemistry
What mass of MgO will be produced when 0.5 Moles of Mg burns in oxygen
Solved
Show answers
More tips
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- C Chemistry 3. Which word best fits with the Earth s day and night cycle a. Revolution b. Rotation C Orbit d. Axis...
- M Mathematics Find two numbers whose difference is 15 and whose product is 1584...
- P Physics Choose the correct words from the box to complete the definition of resistance. current difficulty ease potential difference The electrical resistance of a material is a measure...
- H History Match the correct term to the definition. Column A 1. the dharma, the sangha, and the Buddha: the dharma, the sangha, and the Buddha 2. the Buddha s insights about suffering:...
- C Computers and Technology Register deals with sequencing the execution of instructions. a) stack pointer b) program counter b) accumulator d) flag...
- M Mathematics What s is the volume of a box?...

Ответ:
2Mg + O2 = 2MgO
0,5/2 = х / 2
х = 0,5
0,5* 40 = 20g
Ответ:
Ответ:
Explanation:
pH or pOH is the measure of acidity or alkalinity of a solution.
pH is calculated by taking negative logarithm of hydrogen ion concentration and pOH is calculated by taking negative logarithm of hydroxide ion concentration.
Given: pH= 4.0
Putting in the values in the given equation:
Thus concentration of hydrogen ions [H+] in a solution of pH = 4.0 is