loulou6166
22.04.2020 •
Chemistry
What pressure will be exerted by 0.60 mol of CO2 when its temperature is 25 OC and it is in a 500 mL container?
Solved
Show answers
More tips
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Which compound has the highest percent composition by mass of strontium...
- C Chemistry At room temperature table salt is a solid and mercury is a liquid. what conclusion can you draw about the melting points of these substances?...
- C Chemistry What is the name of the event ,which created the universe ?...
- C Chemistry Shes so pretty i miss her sm...
- C Chemistry Which statement is correct? Use AHxn - 2 (AH(,products) - 2 (AH(resctants). O The enthalpy of the reaction is -184.6 kJ, and the reaction is exothermic. O The enthalpy...
- C Chemistry how much of a Fe(OH) solution with a hydroxide concentration 0.0374 can be made beginning with 3.0 grams?...
- C Chemistry Ava observes that a certain species of bird follows behind a herd of cattle. The bird species primarily eats insects. The birds stay with the herd of cattle as they...
- C Chemistry A bond results from the sharing of valence electrons between atoms. *...
- C Chemistry What are the Δ G ∘ ′ ΔG∘′ values for the component reactions of the creatine kinase reaction? − 43.1 kJ ⋅ mol − 1 −43.1 kJ⋅mol−1 and − 30.5 kJ ⋅ mol − 1 −30.5 kJ⋅mol−1...
- C Chemistry Which two formulas represent Arrhenius acids A)CH3COOH and CH3CH2OH B)HC2H3O2 and KHSO4 C) KHCO3 and KHSO4 D) NaSCN and NA2S2O3...

Ответ:
29.3 atm
Explanation:
We need to use the Ideal Gas Law to solve this problem: PV = nRT, where P is pressure, V is volume, n is moles, R is the gas constant, and T is the temperature in Kelvin.
Here, volume is V = 500 mL = 0.500 L; moles is n = 0.60 mol; R is 0.08206 atm/(mol * K); and temperature is T = 25 + 273 = 298 K. Plug these into the equation to solve for P:
P * (0.500 L) = (0.60 mol) * (0.08206 atm/mol * K) * (298 K)
P ≈ 29.3 atm
Hope this helps!
Ответ:
Explanation:
Relation of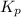 with
with 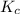 is given by the formula:
is given by the formula:
where,
R = Gas constant
T = temperature
a)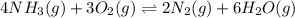
b)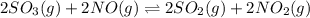
c)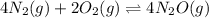
d)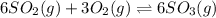
Thus for reaction ,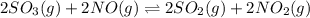 ,
, 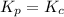 as
as 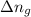 = 0
= 0