angelinamadriga
11.02.2022 •
Chemistry
What type of heat transfer drives plate tectonics?
Solved
Show answers
More tips
- F Food and Cooking How to Make Thin Pancakes: Recipe and Tips...
- S Style and Beauty Is Hot Scissor Haircutting Beneficial or Dangerous?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking Is Bacon Good for You?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Anybody wanna talk ille give brainliest and there are pints...
- P Physics The section of the lever to the right of the fulcrum could be described as the?...
- M Mathematics Kayla had 3/4 of her book left to read on sunday night.she read 1/5 of the book monday afternoon and 1/4 of the book monday evening. what fraction of the book did she still...
- M Mathematics Find the area of the shaded region....
- P Physics In the physics of trajectories, complimentary angles have the same range: for example, projectiles launched from 30 degrees and 60 degrees with the same initial speed will...

Ответ:
Convection, or the flow of mantle material transporting heat, drives plate tectonics.
Explanation:
Have a nice day !!! :)
Ответ:
Answer : The mass of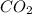 produced form the combustion is, 16.43 kg
produced form the combustion is, 16.43 kg
Explanation :
Density : It is defined as the mass contained per unit volume.
Formula used for density :
First we have to calculate the mass of octane.
Given :
Density of octane = 0.703 g/mL
Volume = 2.00 gallons = 7570 mL
conversion used : 1 gallon = 3785 mL
Now put all the given values in the above formula, we get:
Now we have to calculate the moles of octane.
Molar mass of octane = 114 g/mole
Now we have to calculate the moles of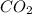 .
.
The balanced chemical combustion reaction of octane will be:
From the balanced chemical reaction we conclude that:
As, 2 moles of react to give 16 moles of
react to give 16 moles of 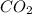
So, 46.68 moles of react to give
react to give 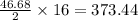 moles of
moles of 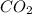
Now we have to calculate the mass of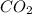 .
.
Molar mass of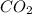 = 44 g/mol
= 44 g/mol
Thus, the mass of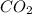 produced form the combustion is, 16.43 kg
produced form the combustion is, 16.43 kg