What volume will be occupied by 11.7 g of Helium in a balloon at an altitude of 10.0 km if the air pressure is 0.262 atm and the temperature is -50.0 °C?
Solved
Show answers
More tips
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- C Chemistry Isotopes name with 13 protons, 12 neutrons and 13 electrons...
- C Chemistry The temperature of the ideal gas in the container shown is 300k. The gas occupies a volume of 1.27. What would be the volume of the gas if it’s temperature were decreased...
- C Chemistry What is the formula for sodium hydrogen phosphate...
- C Chemistry How many particles are in 56 L of nitrogen gas at standard conditions? 0 1.2 x 1023 particles O 2.5 particles O 1.5 x 1024 particles O 2.5 x 1024 particles...
- C Chemistry You may have noticed that there was a big difference in the results of the two hollow pipe have experience but very little difference in solid chunk of experience what...
- C Chemistry Substances that are created and come out of a chemical reaction...
- C Chemistry You dissolve g of succinic acid, , in ml of water. assuming that the density of water is , calculate the molality, mole fraction, and weight percent of acid in the solution....
- C Chemistry What stereochemistry do you expect for the alkene obtained by e2 elimination of (1r,2r)-1,2-dibromo-1,2-diphenylethane? draw a newman projection of the reacting conformation....
- H History Why was life as a nun or missionary appealing to people in the Middle Ages? a. Nuns and monks could marry at that time. b. All nuns and monks were able to visit the...
- M Medicine Elvin decides to make cupcakes for his sister s birthday party. The recipe makes 12 cupcakes, but Elvin needs 24. Elvin decides to double the amount of each measurement...

Ответ:
204 L
Explanation:
According to ideal gas equation:
P = pressure of gas = 0.262 atm
V = Volume of gas = ?
n = number of moles =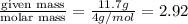
R = gas constant =
T =temperature =
Thus volume occupied by 11.7 g of Helium in a balloon is 204L
Ответ:
mass changes only occur because a gas is being released into the atmosphere, or because a gas from the atmosphere is being used as a reactant - no atoms are being created or destroyed, just rearranged.
Explanation: