Baby010391
22.05.2020 •
Chemistry
What would be the pH of an H2SO4 solution if the [H+] = 4.58 x 10-5 moles/liter?
9.66
4.34
5.67
NEXT QUESTION
3 ASK FOR HELP
Solved
Show answers
More tips
- S Style and Beauty 5 Tips for Choosing the Best Ugg Boots...
- H Health and Medicine Boosting Immunity: A Complete Guide on How to Improve Your Body’s Natural Defenses...
- C Computers and Internet The Best Antivirus Programs for your PC...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- G Geography According to the Knowledge Article, the French were the first to attempt to build a canal across the Isthmus of Panama. However, the French failed in their attempt....
- H History Use your historical sequencing skills. based on the timeline, which roman emperor ruled the longest?...
- B Biology Polydactyly is an autosomal condition. a. dominant b. recessive c. sex-linked d. none of the above...
- P Physics Two particles of masses m and 3m are moving toward each other along the x-axis with the same speed v. they undergo a head- on elastic collision and rebound along the...

Ответ:
pH of an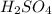 solution is 4.34
solution is 4.34
Explanation:
pH or pOH is the measure of acidity or alkalinity of a solution.
pH is calculated by taking negative logarithm of hydrogen ion concentration.
Putting in the values:
Thus pH of an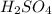 solution if the
solution if the ![[H^+]](/tpl/images/0662/1654/07acb.png) is
is ![4.58\times 10^{-5}]](/tpl/images/0662/1654/335c3.png) is 4.34
is 4.34
Ответ:
x
Explanation: