anabellabenzaquen
03.09.2019 •
Chemistry
When a 0.740-g sample of trinitrotoluene (tnt), c7h5n2o6, is burned in a bomb calorimeter, the temperature increases from 23.4 to 26.9 c the heat capacity of t he calorimeter is 534 j/c, and it contains 675 ml of water. how much heat was produced by the combusttion of the tnt sample?
Solved
Show answers
More tips
- F Food and Cooking How to Determine Healthy, Nutritious Food for Yourself?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Chemistry
- C Chemistry Pls quickly help! Hydrazine (NH4) is often used as a rocket fuel. It can be produced from the elements by reacting nitrogen gas (N2) with another substance. What is the...
- C Chemistry In another experiment, the student titrated 50 ml of .1M HC2H3O2 with .1M NaOH(aq)....
- C Chemistry Why are subscripts used in an ionic bond?...
- H History Are you into straykids ?!...
- M Mathematics Which best describes a three-point thesis? It has three supporting details in the order they will be discussed. It consists of three sentences that form an introductory...
- H Health Yjytnhnyujrrjyryr hynhgh...
- E English Read the sentence. Eliana Rodriguez who teaches biology is my favorite teacher because she gives engaging lectures. Which is the best way to revise this sentence? Eliana...
- E English Here s a public freakout of a kid who doesn t understand what a Brainliest is and thinks I m a beggar...
- S Social Studies How many troops were in the battle of trenton?...
- M Mathematics Someone help me with this ASAP...

Ответ:
Explanation:
The given data is as follows.
mass of sample (X) = 0.740 g, =
= 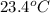
As, Density =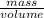
1.00 g/ml =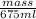
mass = 675 g
Hence, weight of water = 675 g and heat capacity is given as 534
Heat produced by combustion of TNT = Heat gained by the water
Heat =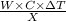
=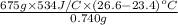
= 1704831 J
As 1 kJ = 1000 J. So, 1704831 J = 1704.831 kJ
Thus, we can conclude that heat produced by the combustion of the TNT sample is 1704.831 kJ.
Ответ:
uh time c
Explanation: