When a chemical reaction occurs,
Solved
Show answers
More tips
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
Answers on questions: Chemistry
- C Chemistry What causes an earthquake?...
- M Mathematics Steve decided to buy a box of pizza from another restaurant. The restaurant is offering a square pizza and a round pizza. Round pizza has an area of 49 pi square inches....
- H History PLZ HELP Based on this map and the Burkhardt photograph, state one way the Cold War affected the city of Berlin. How do we know (aka, what do you see that tells us...
- M Mathematics You are 190 meters from a river. Instead of walking directly towards the river, you walk 440 meters along a straight road to the river s edge. Find the acute angle...
- M Mathematics Find the sides of the angle...
- H History To be human is to be essentially good, despite the capacity for disorder and sin . Do you agree or disagree with this statement. Why or why not?...

Ответ:
Reactions occur when two or more molecules interact and the molecules change. Bonds between atoms are broken and created to form new molecules. That's it.
Explanation:
Ответ:
Answer : The entropy change for the surroundings when 1.74 moles of Na(s) react at standard conditions is
Explanation :
The given balanced chemical reaction is,
First we have to calculate the enthalpy of reaction .
.
where,
We are given:
Putting values in above equation, we get:
Now we have to calculate the entropy change for surrounding .
.
where,
T = temperature = 298 K
Now put all the given values in the above formula, we get:
Now we have to calculate the entropy change for the surroundings when 1.74 moles of Na(s) react at standard conditions.
As, 2 moles of Na(s) has entropy change =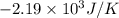
And, 1.74 moles of Na(s) has entropy change =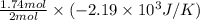
Thus, 23 g of Na(s) has entropy change =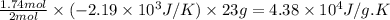
Therefore, the entropy change for the surroundings when 1.74 moles of Na(s) react at standard conditions is