lazavionadams81
04.05.2020 •
Chemistry
When a comet is observed for the first time, astronomers enter information about its position, size, and motion into computer simulations in order to
A.
analyze it for animal life.
B.
measure the length of its tail.
C.
predict its orbital path.
D.
estimate its brightness.
Solved
Show answers
More tips
- H Health and Medicine What makes Graves’ disease dangerous?...
- W Work and Career 10 Best Ways To Find A Job: Tips To Land Your Dream Job...
- L Leisure and Entertainment How to Choose a Program for Cutting Music?...
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- M Mathematics Function a and function b are linear functions. function a isrepresented by the table of values function b is represented by the equation. function a table: x. y 1. 2...
- M Mathematics Find the coordinates of the circumcenter of triangle pqr with vertices p(-2,5), q(4,1), and r(-2,-3). the coordinates of the circumcenter are ( , )....
- M Mathematics This diagram illustrates an unfolded part of a greeting card, What is its area ?...
- P Physics What is the mass of an object that has a density of 1.3 g/cm^3 and a volume of 2cm^3...

Ответ:
The unit cell edge length for the alloy is 0.405 nm
Explanation:
Given;
concentration of Ag,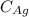 = 78 wt%
= 78 wt%
concentration of Pd, = 22 wt%
= 22 wt%
density of Ag = 10.49 g/cm³
density of Pd = 12.02 g/cm³
atomic weight of Ag,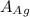 = 107.87 g/mol
= 107.87 g/mol
atomic weight of iron,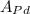 = 106.4 g/mol
= 106.4 g/mol
Step 1: determine the average density of the alloy
Step 2: determine the average atomic weight of the alloy
Step 3: determine unit cell volume
for a FCC crystal structure, there are 4 atoms per unit cell; n = 4
Step 4: determine the unit cell edge length
Vc = a³
Therefore, the unit cell edge length for the alloy is 0.405 nm