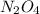nails4life324
03.06.2021 •
Chemistry
When a very massive star runs out of elements to fuse, it
Solved
Show answers
More tips
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- P Philosophy How to Develop Extrasensory Abilities?...
Answers on questions: Chemistry
- C Chemistry Explain why the anion solutions were prepared using sodium salts rather than calcium salts?...
- C Chemistry Suppose 68.9 g of hexane is mixed with 75. g of oxygen. Calculate the minimum mass of hexane that could be left over by the chemical reaction. Be sure your answer has the...
- M Mathematics A rectangular prism with a volume of 10 cubic units is filled with cubes with side lengths of 1/2...
- M Mathematics ( - 7a y + xy + 3x + 2) + (5r²y – 5cy - 6x – 7)...

Ответ:
Empirical formula is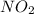 and molecular formula is
and molecular formula is 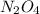
Explanation: To find the empirical and molecular formula, we follow few steps:
Step 1: Converting mass percent into mass
We are given the percentage of elements by mass. So, the total mass take will be 100 grams.
Therefore, mass of nitrogen =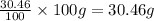
Similarly, mass of oxygen =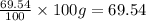
Step 2: Converting the masses into their respective moles
We use the formula:
Molar mass of Nitrogen = 14 g/mol
Molar mass of oxygen = 16 g/mol
Moles of nitrogen =
Moles of oxygen =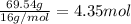
Step 3: Getting the mole ratio of nitrogen and oxygen by dividing the calculated moles by the lowest mole value.
Mole ratio of nitrogen =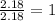
Mole ratio of oxyegn =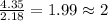
Step 4: The mole ratio of elements are represented as the subscripts in a empirical formula, we get
Empirical formula =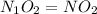
Step 5: For molecular formula, we divide the molar mass of the compound by the empirical molar mass.
Empirical molar mass of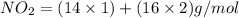
Empirical molar mass = 46 g/mol
Molar mass of the compound = 92 g/mol
Now, multiplying each of the subscript of empirical formula by 'n', we get
Molecular formula =