savthespice
10.09.2019 •
Chemistry
When m2s3(s) is heated in air, it is converted to mo2(s). a 3.280 g sample of m2s3(s) shows a decrease in mass of 0.228 g when it is heated in air. what is the average atomic mass of m?
Solved
Show answers
More tips
- C Computers and Internet How to Create a Website for Free and Easy?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry The amount of energy released when one or more electron is added to the neutral atom ▪︎electron affinity▪︎electronegativity...
- C Computers and Technology Movie recommendations?...
- S SAT El proyecto para proteger el medio ambiente 1 of 1 se canceló la semana pasada....
- M Mathematics Two legs of a right triangle are 2 inches and 3 inches long. What is the length of the shorter leg of a similar right triangle if its longer leg is 12 inches long PLease Need your...
- M Mathematics This is algebra 2 naming polynomials can some plzzz help me I am struggling...
- S Social Studies Salient features of sociology...

Ответ:
The average atomic mass of M is 181.33 g/mol.
Explanation:
First off we need to know the reaction that takes place. The balanced reaction of M₂S₃(s) is:
M₂S₃(s) + O₂(g) ⇒2MO₂(s) + 3SO₂(g)The important section for this problem is this:
M₂S₃(s) ⇒2MO₂(s)**Thus, the number of moles of M in M₂S₃ is equal to the number of moles of M in 2MO₂.
The decrease in mass means that M₂S₃ reacted and produced MO₂, thus the mass of MO₂ is 3.280-0.228=3.052 g
Now let's say x is the atomic weight of M, and write the molecular weights (Mw) of those two compounds:
Mw of M₂S₃= 2x + 96 g/mol
Mw of MO₂= x + 32 g/mol
Now we determine the moles of each compound, using the formula [ moles = mass / molecular weight ]:
Moles of M₂S₃=
Moles of MO₂=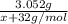
Using the equivalence marked by asterisks, we're left with (note that the second denominator is multiplied by 2 because of the reaction coefficients):
We solve for x:
Thus, the average atomic mass of M is 181.33 g/mol.
Ответ:
The water molecule has a total of 10 protons and 10 electrons (8 from the oxygen atom and 1 from each of the two hydrogen atoms). Since it has the same number of protons and electrons, the water molecule is neutral. The electron cloud model shows where electrons are in a molecule.
- BRAINLIEST answerer