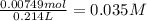makayyafreeman
10.06.2021 •
Chemistry
When the nuclide polonium-218 undergoes alpha decay:
a. The name of the product nuclide is .
b. The symbol for the product nuclide is .
Write a balanced nuclear equation for the following: The nuclide polonium-218 undergoes alpha emission.
Solved
Show answers
More tips
- P Philosophy What is Something for you?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry The density of aluminum is 2.70g/cm^. what is the mass of a solid piece of aluminum with a volume of 1.50 cm^3?...
- C Chemistry According to the following reaction, how many grams of hydrofluoric acid will be formed upon the complete reaction of 25.6 grams of water with excess silicon tetrafluoride? silicon...
- C Chemistry Convert 630100 to scientific notation...
- C Chemistry The fission of a nucleus is accompanied by the release of very little energy. true or false...
- C Chemistry Aradioactive alkaline earth element’s atomic number increases by two after emitting alpha radiation. a. alpha, beta, gamma b. alpha, gamma, beta c. x-ray, beta, gamma d. x-ray, gamma,...
- C Chemistry the mystery of periodic law was solved when proposed his planetary atomic model of the atom, providing an understanding of the electronic structure of the elements and the organization...
- C Chemistry Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of...
- C Chemistry Areaction is first order. if the initial reactant concentration is 0.0200 m, and 25.0 days later the concentration is 6.25 x 10-4 m, then its half-life is:...
- E English Japanese haiku are similar to english romantic poems because they both...
- M Mathematics Me answer this maths question.☺***must show step by step guide to how you got your answers***☺...

Ответ:
a): The name of the product nuclide is lead-214
b): The symbol of the product nuclide is Pb-218
Explanation:
There are three types of decay processes:
Alpha decayBeta decayGamma decayAlpha decay is the decay process that happens when a heavy nucleus decays into a light nucleus with the release of an alpha particle. This alpha particle carries a charge of +2 units and has a mass of 4 units. It is also known as the helium nucleus. The general equation for this decay process is:
The nuclear equation for the alpha decay of Po-218 follows:
Hence, the name of the product nuclide is lead-214 and the symbol is Pb-218.
Ответ:
a.3.2204 g
b.Ag+ = 0 M
NO3? = 0.105 M
Ca2+ = 0.07M
Cl? =0.035 M
Explanation:
The balanced equation for this reaction is:
mol of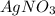
mol of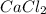
from the reaction stoichiometry we know that we need 2 moles of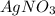 per every mol of
per every mol of 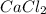 .
.
so for 0.01498 mol of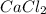 we need 0.01498*2 =0.02996 mol of
we need 0.01498*2 =0.02996 mol of 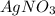 . Since we have just 0.02247 mol, this will be the reaction limit reagent for this reaction.
. Since we have just 0.02247 mol, this will be the reaction limit reagent for this reaction.
we will use all the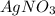
mol of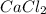 used =
used =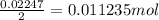
For the calculation we always use the limit reagent.
a. How many grams of silver chloride.
From the reaction stoichiometry we know that 2 mol of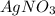 produce 2 mol of AgCl so, 0.02247 mol of
produce 2 mol of AgCl so, 0.02247 mol of 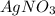 will the same number of mol of AgCl.
will the same number of mol of AgCl.
AgCl produced = 0.02247 mol
molecular weight of AgCl = 143.32 g/mol
mass of AgCl =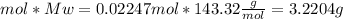
b.concentrations of each ion
volume of the remained solution 107 mL + 107mL= 214 mL or 0.214 L
concentration =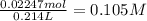
following the same analysis for made for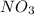 we know that
we know that 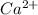 did not precipitated so we have the same number of moles of
did not precipitated so we have the same number of moles of 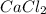 in the original sample but in a different volume
in the original sample but in a different volume
concentration =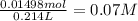
the Cl in the solution after the precipitation will be the Cl that did not reacted since all the Cl that reacted to produce AgCl precipitated.
Cl that reacted is the same number of AgCl produced this is=0.02247 mol
Cl before the reaction is 2 times the number of moles of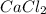 in the sample this is: 0.01498*2 =0.02996 mol
in the sample this is: 0.01498*2 =0.02996 mol
Cl remained = 0.02996 mol -0.02247 mol=0.00749 mol
concentration =