Which is four products of a reaction type?
Solved
Show answers
More tips
- W Work and Career Secrets of Punctuality: How to Learn to Never Be Late?...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- C Chemistry Please I need an explanation on the relationship between concentration and volume of reacting substances...
- C Chemistry Trình bày, giải thích cấu tạo của hạt mixen keo dựa trên quan điểm của thuyết Stern ?...
- C Chemistry Which of the following when dissolved in deionized water would make a good conducting solution? a)methanolb)sugarc)baking soda...
- C Chemistry What is the molarity (m) of a solution with 2 mol of nacl in 0.50 l of solution?...
- C Chemistry * Required 1. The diagram below shows the repeating groups of atoms that make up two samples. Will the properties of the two samples likely be the same or different? (Examples...
- C Chemistry What else should I add for creativity? Or else I won’t receive credit...
- C Chemistry The of an adult male is 750 newtons....
- C Chemistry How has the work of chemists affected the environment over the years...
- C Chemistry What is the total number of moles of Ca(NO3)2 needed to make 7.3 liters of a 5.1 M Ca(NO3)2 solution? Please help quick...
- C Chemistry What is the mass of 2.40 mol H2O? Answer in units of g....

Ответ:
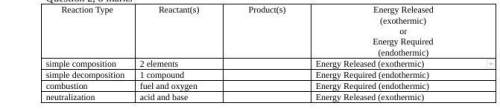
1=2H₂ + O₂ → 2H₂O
2=CaCo₃ + heat → CaO +CO₂
3=CH₄ + 2O₂ → CO₂ +2H₂O
4=HCl + NaOH → NaCl + H₂O
Explanation:
1 = Simple composition
The formation of water molecule is simple composition reaction. In this reaction two hydrogen atoms react with one oxygen atom and form one water molecules.
2H₂ + O₂ → 2H₂O
The amount of energy released is -285.83 KJ/mol. It is exothermic reaction.
2 = Simple decomposition reaction:
The break down of sodium hydrogen carbonate into sodium carbonate, carbondioxide and water is decomposition reaction. The decomposition reactions re mostly endothermic, because compound required energy to break.
2NaHCO₃ + heat → Na₂CO₃ + H₂O + CO₂
It is endothermic reaction.
Another example is:
CaCo₃ + heat → CaO +CO₂
3 = Combustion reaction
Consider the combustion of methane:
CH₄ + 2O₂ → CO₂ +2H₂O
The burning of methane is exothermic. The combustion reactions are exothermic because when fuel are burns they gives energy.
4 = Neutralization reaction
The neutralization reactions are those in which acid and base react to form the salt and the water. Some neutralization reactions are exothermic because they release heat. e.g
Consider the neutralization reaction of HCl and NaOH.
HCl + NaOH → NaCl + H₂O
Ответ:
It’s inappropriate question and it was not even a question