totallybriii
07.01.2020 •
Chemistry
Which is the most ethical response if an experiment fails to confirm a hypothesis?
question 2 options:
revise the hypothesis and develop a new experiment to test it.
alter the data so that they conform to what we already know.
perform the experiment again until it confirms the hypothesis.
publish the data without any conclusions.
Solved
Show answers
More tips
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- D Dating, Love, Relationships How Long Can Love Last?...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- F Food and Cooking How to Quickly Put your Child to Sleep?...
- C Computers and Internet How to Create a Website for Free and Easy?...

Ответ:
Ответ:
The specific heat capacity of the unknown metal is 1.04 J/g°C.
Explanation:
Let the specific heat capacity of the unknown metal be .
.
Given:
Mass of the metal is,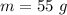
Initial temperature of the metal is,
Volume of water is,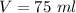
Specific heat capacity of water is,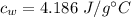
Initial temperature of water is,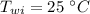
Final temperature of the system is,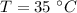
We know that density of water is equal to 1 g/ml.
Mass is given as the product of density and volume.
Therefore, mass of water is given as:
Now, fall of temperature of the unknown metal is given as:
Rise of temperature of water is given as:
Now, as per conservation of energy,
Heat lost by metal = Heat gained by water
⇒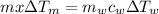
Plug in all the given values and solve for . This gives,
. This gives,
Therefore, the specific heat capacity of the unknown metal is 1.04 J/g°C.