11needhelp11
29.06.2019 •
Chemistry
Which kind of nuclear decay does not alter the identity of the atom?
Solved
Show answers
More tips
- W Work and Career Which types of activities are not subject to mandatory licensing?...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry How many atoms are in 5.76 moles of aluminum?...
- H History What are the egypt pyramids made out of ?...
- B Business All of the following theories show why it is beneficial for a country to engage in international trade even for products it is able to produce for itself, except:. 1. Comparative...
- S Social Studies Is rule by few. these people could hold all the power based on military strength, family power, or religious force. a. oligarchy b. theocracy c. democracy d. monarchy...
- M Mathematics What is the sum of one gross, a quarter of a dozen and two scores...

Ответ:
gamma decay
Explanation:
1. Alpha decay: In this process, alpha particles is emitted when a heavier nuclei decays into lighter nuclei. The alpha particle released has a charge of +2 units.
2. Beta-decay: In this process, a neutron gets converted into a proton and an electron releasing a beta-particle. The beta particle released carries a charge of -1 units.
3. Gamma decay: in this process, an unstable nuclei gives off excess energy by a spontaneous electromagnetic process and releases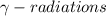 . These radiations does not carry any charge and are electrically neutral and thus the identity of the atom undergoing gamma decay do not change.
. These radiations does not carry any charge and are electrically neutral and thus the identity of the atom undergoing gamma decay do not change.
Ответ:
At equilibrium state, the water will have turned into ice.
Explanation:
An Equilibrium phase is defined as a stage when chemical potential of any component ( solid liquid or gas) present in the system remains steady with time. So, in this situation, because the system is perfectly insulated, no heat will be gained or lost. The water and ice are both at exactly 0 °C, then that temperature will remain constant. As time goes on, the water will freeze and the system will become all ice.