laurydamas2002
21.08.2019 •
Chemistry
Which of the following generally results when the senate and the house of representatives pass different versions of the same bill?
Solved
Show answers
More tips
- L Leisure and Entertainment How to Learn to Draw Graffiti: Tips for Beginners...
- P Photography and Videography How to Choose a Digital Camera?...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- M Mathematics In the figure shown, line AB is parallel to line CD. What is the measure of angle x? Explain your answer. А P B 62° 115° с о R 0 (10 points) HTML Editor...
- H History Civic obligations of american citizens pay taxes agree to serve on a jury if selected register, if required, for the military selective service which item completes the...

Ответ:
Ответ:
Complete Question
The rate of a certain reaction is given by the following rate law:
rate Use this information to answer the questions below.
What is the reaction order in ?
?
What is the reaction order in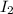 ?
?
What is overall reaction order?
At a certain concentration of H2 and I2, the initial rate of reaction is 2.0 x 104 M / s. What would the initial rate of the reaction be if the concentration of H2 were doubled? Round your answer to significant digits. The rate of the reaction is measured to be 52.0 M / s when [H2] = 1.8 M and [I2] = 0.82 M. Calculate the value of the rate constant. Round your answer to significant digits.
The reaction order in is n = 1
is n = 1
The reaction order in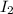 is m = 1
is m = 1
The overall reaction order z = 2
When the hydrogen is double the the initial rate is
The rate constant is
Explanation:
From the question we are told that
The rate law is![rate = k [H_2][I_2]](/tpl/images/0667/2967/0cf2e.png)
The rate of reaction is
Let the reaction order for be n and for
be n and for 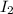 be m
be m
From the given rate law the concentration of is raised to the power of 1 and this is same with
is raised to the power of 1 and this is same with 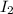 so their reaction order is n=m=1
so their reaction order is n=m=1
The overall reaction order is
At
= >![k = \frac{2.0*10^{4}}{[H_2] [I_2] }](/tpl/images/0667/2967/c78a9.png)
given that the concentration of hydrogen is doubled we have that
=>![k = \frac{rate_n }{ [2H_2] [I_2]}](/tpl/images/0667/2967/3b211.png)
So equating the two k
=>
So when
We have