theflash077
10.03.2021 •
Chemistry
Which of the following is an electrolyte?
a. BaSO4
b. CaS
c. AlPO4
d. SrCrO4
Solved
Show answers
More tips
- A Animals and plants Уход за джунгариками: полезные советы и рекомендации...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- M Mathematics Aroad map has a scale of 1: 20000 on the map a road has a length of 4cm work out the actual distance five your answer in metre...
- C Chemistry Cesium (cs) has one valence electron, and that electron is located in the sixth subshell. without using the periodic table, in which group and period is cesium located? group...
- M Mathematics GEOMETRY - triangle similarity...
- S Social Studies Why was the Niger River important to the growth of the Mali Empire? It protected the Mali Empire from invasion. It provided fertile farm soil and trade routes. It served as...

Ответ:
Answer is in the photo. I can't attach it here, but I've uploaded it to a file hosting. link below! Good Luck!
cutt.ly/Kzv78uJ
Ответ:
A
Explanation:
Ответ:
a negative ion that attracts a proton from water is the correct answer.
Explanation:
An acidic solution is defined as a solution that is formed when a strong acid combines with a weak base.
For example,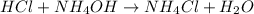
So, when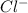 from
from 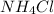 attract a proton from water then it will form HCl which is acidic in nature and being a strong acid will result in an acidic solution.
attract a proton from water then it will form HCl which is acidic in nature and being a strong acid will result in an acidic solution.
When a solution is formed from a strong bases and weak acid then it will result in a basic solution. When a weak acid will react with a weak base then it will result in a neutral solution.
Hence, we can conclude that a negative ion that attracts a proton from water is the kind of ion that is contained in salts that produce an acidic solution.