shiyaharper
23.10.2020 •
Chemistry
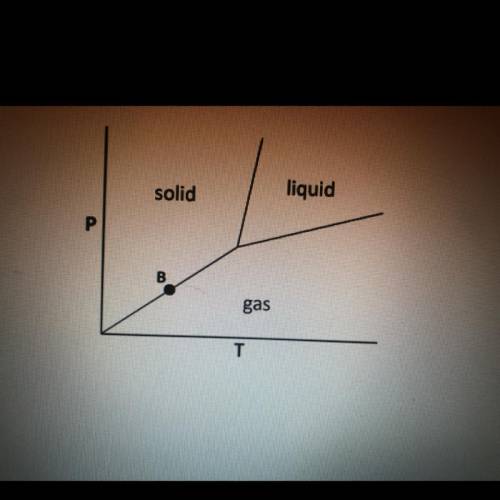
Which of the following phase occurs at point b on the phase diagram shown below?
A) deposition
B)freezing
C)melting
D) boiling
Solved
Show answers
More tips
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- S Social Studies Which emperor made christianity the official religion of the roman empire?...
- H History How many ways can two marbles be chosen from a set of five marbles? 5 10 15 20...
- B Business Arep at the firm has been working on determining appropriate investments for a client. the rep comes to the conclusion that a deferred variable annuity will be the best...
- P Physics Describe the movement of a transverse wave please i will give brainliest and the max points...

Ответ:
I think it’s b.
Explanation:
Ответ:
= 5.4×10-6 g AgBr
Explanation;AgBr(s) → Ag+(aq) + Br-(aq)Ksp = 3.3×10^-13
[Ag+][Br-] = Ksp = 3.3×10^-13 = X²
X = 5.7×10^-7 M = [Ag+] = mol/L AgBr that dissolve
1 mole of AgBr = 187.8 g
Therefore;
= 0.05 L × (5.7 × 10^-7 mol/L) × (187.8 g /mol)
= 5.4×10-6 g AgBr
2. Answer and explanation;The two factors that accounts for increased rate of chemical reaction when temperature is increased are:
Energy factor; enough energy in the collision for the formation of an activated complex, where bonds are breaking and new ones forming. When temperature is increased, a greater number of molecular collisions possess enough energy to activate the reaction.Frequency of collisions increases; an increase in temperature makes particles move faster and collide more frequently, increasing the possibility of a reaction be-tween them.More important factor; Energy factor is more important.