HistoryLee
29.06.2019 •
Chemistry
Which of these is an example of a chemical change a: fireworks exploding b: grinding powder c: melting cheese d: ironing shirt
Solved
Show answers
More tips
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry What are the reactants found in soap and detergents?...
- C Chemistry Transition metals tend to a. form cations b. lose electrons from the ns subshell first c. form more than one ion d. all of the above...
- C Chemistry Astudent uses 0.10 m hcl to simulate the acid concentration in the stomach. what volume( in ml) of “stomach acid” reacts with a tablet that contains 0.10 g of magnesium...
- C Chemistry Need plz i am confused on what to do! instructions: read each myth (untruth). reword it to make a factual statement. then, give two to three reasons why the myth is untrue....
- C Chemistry Kinetic or potential energy....
- C Chemistry When a sample of gas is heated, its thermal energy...
- C Chemistry A scientist measures out precisely 1.45 mg of a chemical. How many microg is this equal to...
- C Chemistry How do you calculate the mass of a sample of gold which has a volume of 4.35 mL and a density of 19.3 g/mL...
- C Chemistry HELPPP 1. A solution containing 70 g of NaNO3 at 30 ° C (in 100 ml of H2O) is it saturated or insaturated 2. A solution containing 50 g of NH4CI at 50 ° C (in 100 ml of...
- M Mathematics I need to know the answers...

Ответ:
The chemical change is A: Fireworks Exploding
Ответ:
the answer is a FIREWORKS plz mark me brainliest
Ответ:
0.35 milli moles of ethanol can be theoretically be produced under these conditions.
Explanation:
Moles of glucose =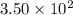 milli mole
milli mole
Moles of ADP = 0.35 milli mole
Moles of Pi = 0.35 milli mole
Moles of ATP = 0.70 milli mole
As we can see that ADP and Pi are in limiting amount which means tat they are limiting reagent. So, the moles of ethanol produced will depend upon the moles of ADP and Pi.
According to reaction, 2 moles of ADP gives 2 moles of glucose.
Then 0.35 milli moles of ADp will give :
0.35 milli moles of ethanol can be theoretically be produced under these conditions.