Which of these statements are true? select all that apply.
the hrxn for the reaction c(s) + o2(g) co2(g) is the same as the hf for co2(g). the hrxn for the reaction na(s) + cl2(g) 2nacl(s) is the same as the hf for nacl(s). the hf for br2(l) is 0 kj/mol by definition.
to determine the hf for h2o(g), just reverse the sign of the hrxn for the reaction 2h2o(l) 2h2(g) + o2(g).
the hf for n2(l) is 0 kj/mol by definition.
the hrxn for the reaction 1.5h2(g) + 0.5n2(g) nh3(g) is the same as the hf for nh3(g).
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking Is Bacon Good for You?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- M Mathematics Please Help!? This is due tomorrow...
- M Mathematics A scale drawing of a rectangular park had a scale of 1 cm = 120 m. 5.4 cm -11.1 cm What is the actual area of the park in meters squared?...
- M Mathematics Lovell recorded the number of tickets and hot dogs sold at King Stadium. He listed the sales in the table below. Tickets Sold Hot Dogs Sold KING STADIUM SALES DATA 3781...

Ответ:
Enthalpy of formation:
The change in enthalpy when one mole of substance is formed from its constituent elemetns at standard state.
Enthalpy of reaction:
The change in enthalpy when a reaction occurs and the reactants and products are in their standard states.
Now, we check the statements. The true ones are:
The Hrxn for C(s) + O₂(g) → CO₂(g) is the same as Hf for CO₂
This is true because the formation of carbon dioxide requires carbon and oxygen in their standard states.
The Hf for Br₂(l) is 0 kJ/mol by definition.
Because the bromine is present in its standard state, the enthalpy of formation is 0.
The Hrxn for the reaction 1.5H₂(g) + 0.5N₂(g) → NH₃(g) is the same as the Hf for NH₃(g)
The reactants and products are present in their standard state, and the reaction is the same as the one occurring during the formation of ammonia.
Ответ:
The statements that are true are 1, 3, and 6. The statements are:
TheThe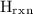 has been a change in the enthalpy when the reaction takes place. The
has been a change in the enthalpy when the reaction takes place. The  has been the enthalpy of formation of a product in the reaction.
has been the enthalpy of formation of a product in the reaction.
1. The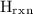 for the reaction C(s) +
for the reaction C(s) +  (g)
(g) 
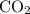 (g) is the same as the
(g) is the same as the  for
for 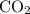 (g).
(g).
The statement has been true, as the formation of gaseous carbon dioxide requires the carbon and oxygen to be present in their standard existing states.
2. The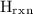 for the reaction Na(s) + Cl2(g) 2NaCl(s) is the same as the
for the reaction Na(s) + Cl2(g) 2NaCl(s) is the same as the  for NaCl(s).
for NaCl(s).
The reaction has been required with the presence of Cl in the aqueous state, and the formed NaCl will be liquid. Thus, the statement has been false.
3. The for Br2(l) is 0 kJ/mol by definition.
for Br2(l) is 0 kJ/mol by definition.
The bromine has been existing in a liquid state in nature, thus the enthalpy of formation of the liquid has been zero. Thus the statement is true.
4. To determine the for
for  (g), just reverse the sign of the
(g), just reverse the sign of the 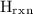 for the reaction
for the reaction 
The state of hydrogen and oxygen has been different for the formation, thus the statement is false.
5. The for
for 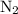 (l) is 0 kJ/mol by definition.
(l) is 0 kJ/mol by definition.
The natural existing state of nitrogen is gaseous. Thus the statement has been false.
6. The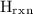 for the reaction
for the reaction ) is the same as the
) is the same as the  for
for  .
.
The statement has been true because of the same states of reactants for the formation of ammonia.
Thus the statements that are true are 1, 3, and 6.
For more information about the enthalpy of formation, refer to the link:
link
Ответ:
(Answer) The name of the compound with formula N2Cl4 is dinitrogen tetrachloride.
In this compound, two central nitrogen atoms are bonded by covalent bonds by sharing valence electron pairs among each other. Two chlorine atoms are attached to each nitrogen atom by covalent bond formation.
One molecule of the compound N2Cl4 has 2 nitrogen atoms and 4 chlorine atoms. For this reason, its name is dinitrogen tetrachloride. Here, "di" represents "two" and "tetra" represents "four".