maddyclark19
22.07.2019 •
Chemistry
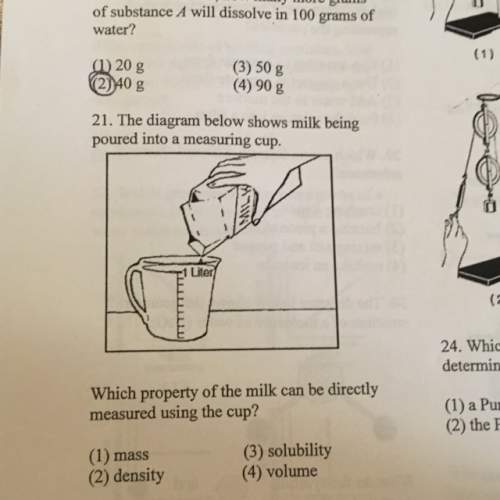
Which property of the milk can be directly measured using the cup?
Solved
Show answers
More tips
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- M Mathematics Please, I need help! The table above shows the number of trees and the number of apples that Jacob picked from each tree. Answer the following questions based on he table above...
- S Spanish Which of the following commands is not in the negative tú form?...
- B Biology Keeping mitosis and meiosis separate in your mind can be a bit tricky. read the following speech bubbles. determine whether they would be correctly placed in the mitosis clip,...

Ответ:
Ответ:
9 moles of NaNO₃.
Explanation:
We'll begin by writing the balanced equation for the reaction. This is illustrated below:
Al(NO₃)₃ + 3NaCl –> 3NaNO₃ + AlCl₃
From the balanced equation above,
1 mole of Al(NO₃)₃ reacted with 3 moles of NaCl to produce 3 moles of NaNO₃.
Next, we shall determine the limiting reactant. This can be obtained as follow:
From the balanced equation above,
1 mole of Al(NO₃)₃ reacted with 3 moles of NaCl.
Therefore, 4 moles of Al(NO₃)₃ will react with = 4 × 3 = 12 moles of NaCl.
From the calculation made above, we can see that it will take a higher amount (i.e 12 moles) of NaCl than what was given (i.e 9 moles) to react completely with 4 moles of Al(NO₃)₃. Therefore, NaCl is the limiting reactant and Al(NO₃)₃ is the excess reactant.
Finally, we shall determine the maximum amount of NaNO₃ produced from the reaction.
In this case, the limiting reactant will be use because it will produce the maximum amount of NaNO₃ since all of it is used up in the reaction.
The limiting reactant is NaCl and maximum amount of NaNO₃ produced can be obtained as follow:
From the balanced equation above,
3 moles of NaCl reacted to produce 3 moles of NaNO₃.
Therefore, 9 moles of NaCl will also react to produce 9 moles of NaNO₃
Thus, 9 moles of NaNO₃ were obtained from the reaction.